

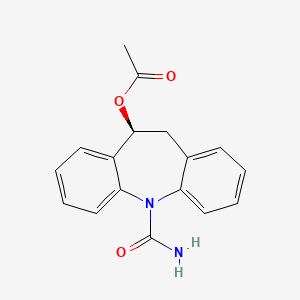

1. 10-acetoxy-10,11-dihydro-5h-dibenz(b,f)azepine-5-carboxamide
2. Aptiom
3. Bia 2-093
4. Bia-2-093
5. Zebinix
1. 236395-14-5
2. Bia 2-093
3. Zebinix
4. Exalief
5. Aptiom
6. Stedesa
7. Erelib
8. Pazzul
9. Bia-2-093
10. Eslicarbazepine (acetate)
11. (s)-5-carbamoyl-10,11-dihydro-5h-dibenzo[b,f]azepin-10-yl Acetate
12. Sep-0002093
13. Eslicarbazepine Acetate [usan]
14. Bea68zvb2k
15. Chembl87992
16. Sep - 0002093
17. Chebi:87016
18. [(5s)-11-carbamoyl-5,6-dihydrobenzo[b][1]benzazepin-5-yl] Acetate
19. Bia-2093
20. Exelief
21. (s)-(-)-10-acetoxy-10,11-dihydro-5h-dibenz[b,f]azepine-5-carboxamide
22. S-(-)-10-acetoxy-10,11-dihydro-5h-dibenz[b,f]azepine-5-carboxamide
23. Eslicarbazepine Acetate (usan)
24. (10s)-10-acetoxy-10,11-dihydro-5h-dibenz[b,f]azepine-5-carboxamide
25. (10s)-5-carbamoyl-10,11-dihydro-5h-dibenzo[b,f]azepin-10-yl Acetate
26. (s)-10-acetoxy- 10,11-dihydro- 5h-dibenz[b,f]azepine- 5-carboxamide
27. 5h-dibenz[b,f]azepine-5-carboxamide, 10-(acetyloxy)-10,11-dihydro-, (10s)-
28. Sep 0002093
29. Unii-bea68zvb2k
30. (10s)-5-carbamoyl-10,11-dihydro-5h-dibenzo(b,f)azepin-10-yl Acetate
31. 5h-dibenz(b,f)azepine-5-carboxamide, 10-(acetyloxy)-10,11-dihydro-, (10s)-
32. Bia 2093
33. Zebinix (tn)
34. Aptiom (tn)
35. Eslicarbazepine-acetate
36. Eslicarbazapine Acetate
37. Schembl250594
38. Zinc7295
39. Dtxsid90178308
40. Hms3263m06
41. Hms3885n14
42. Hy-b0703
43. Eslicarbazepine Acetate [mi]
44. Tox21_501102
45. Bdbm50240669
46. Mfcd06798333
47. S4657
48. Akos005145781
49. Ccg-222406
50. Db09119
51. Eslicarbazepine Acetate [mart.]
52. Eslicarbazepine Acetate [vandf]
53. Lp01102
54. Sdccgsbi-0633789.p001
55. Eslicarbazepine Acetate [who-dd]
56. Ncgc00165752-01
57. Ncgc00165752-02
58. Ncgc00165752-04
59. Ncgc00165752-06
60. Ncgc00261787-01
61. Ac-31734
62. As-14058
63. Eslicarbazepine Acetate [ema Epar]
64. E1046
65. Eslicarbazepine Acetate [orange Book]
66. Bia 2-093, >=98% (hplc), Solid
67. D09612
68. H11471
69. Ar-270/43507894
70. Q410273
71. J-015175
72. (s)-10-acetoxy-10,11-dihydro-5hdibenz( B,f)azepine-5-carboxamide
73. 10s)-5-carbamoyl-10,11-dihydro-5h-dibenzo[b,f]azepin-10-yl Acetate
74. 5-(aminocarbonyl)-10,11-dihydro-5h-dibenzo[b,f]azepin-10-yl Acetate
75. 5h-dibenz9b,f)azepine-5-carboxamide, 10-(acetyloxy)-10,11-dihydro-, (10s)-
76. Acetic Acid (s)-5-carbamoyl-10,11-dihydro-5h-dibenzo[b,f]azepin-10-yl Ester
77. Acetic Acid 5-carbamoyl-10,11-dihydro-5h-dibenzo[b,f]azepin-10(s)-yl Ester
| Molecular Weight | 296.32 g/mol |
|---|---|
| Molecular Formula | C17H16N2O3 |
| XLogP3 | 2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Exact Mass | 296.11609238 g/mol |
| Monoisotopic Mass | 296.11609238 g/mol |
| Topological Polar Surface Area | 72.6 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 440 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Aptiom |
| PubMed Health | Eslicarbazepine (By mouth) |
| Drug Classes | Anticonvulsant, Central Nervous System Agent |
| Drug Label | The chemical name of APTIOM (eslicarbazepine acetate) is (S)-10-Acetoxy-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide. APTIOM is a dibenz[b,f]azepine-5-carboxamide derivative. Its molecular formula is C17H16N2O3 and its molecular weight is 296.32... |
| Active Ingredient | Eslicarbazepine acetate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 600mg; 800mg; 400mg |
| Market Status | Prescription |
| Company | Sunovion Pharms |
| 2 of 2 | |
|---|---|
| Drug Name | Aptiom |
| PubMed Health | Eslicarbazepine (By mouth) |
| Drug Classes | Anticonvulsant, Central Nervous System Agent |
| Drug Label | The chemical name of APTIOM (eslicarbazepine acetate) is (S)-10-Acetoxy-10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide. APTIOM is a dibenz[b,f]azepine-5-carboxamide derivative. Its molecular formula is C17H16N2O3 and its molecular weight is 296.32... |
| Active Ingredient | Eslicarbazepine acetate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 600mg; 800mg; 400mg |
| Market Status | Prescription |
| Company | Sunovion Pharms |
Eslicarbazepine acetate is indicated as adjunctive therapy in the treatment of partial-onset seizures that are not adequately controlled with conventional therapy in epileptic patients.
FDA Label
Zebinix is indicated as adjunctive therapy in adults, adolescents and children aged above 6 years, with partial-onset seizures with or without secondary generalisation.
Exalief is indicated as adjunctive therapy in adults with partial-onset seizures with or without secondary generalisation.
Treatment of epilepsy with partial-onset seizures
Eslicarbazepine acetate is associated with a dose- and concentration-dependant increase in heart rate and prolongation of PR interval.
Anticonvulsants
Drugs used to prevent SEIZURES or reduce their severity. (See all compounds classified as Anticonvulsants.)
Voltage-Gated Sodium Channel Blockers
A class of drugs that inhibit the activation of VOLTAGE-GATED SODIUM CHANNELS. (See all compounds classified as Voltage-Gated Sodium Channel Blockers.)
N03AF04
N03AF04
Absorption
Eslicarbazepine active metabolite has a high bioavailability and reaches peak serum concentration 1-4 hours after a given dose. Eslicarbazepine acetate absorption is not affected by food.
Route of Elimination
Eslicarbazepine acetate and its metabolites are eliminated primarily via renal excretion. Eslicarbazepine active metabolite is excreted two-thirds in the unchanged form and one-third as a glucuronide conjugate. This accounts for around 90% of total metabolites excreted, with the remaining 10% being minor metabolites. Renal tubular reabsorption is expected to occur with eslicarbazepine.
Volume of Distribution
The apparent volume of distribution of eslicarbazepine is 61.3 L for a body weight of 70 kg based on population PK analysis.
Clearance
Renal clearance of eslicarbazepine was found to be approximately 20 mL/min in healthy subjects with normal renal function.
Eslicarbazepine acetate is rapidly and extensively metabolized to its major active metabolite, eslicarbazepine, via hydrolytic first-pass metabolism. Eslicarbazepine corresponds to about 92% of systemic exposure. Minor active metabolites (R)-licarbazepine and oxcarbazepine consist of <5% of systemic exposure. Active metabolites are then metabolized to inactive glucuronides that correspond to about 3% of systemic exposure. Eslicarbazepine had a moderate inhibitory effect on CYP2C19 and a mild activation of UGT1A1-mediated glucuronidation when studied in human hepatic microsomes. It has been shown to induce CYP3A4 enzymes in vivo.
The apparent plasma half-life of eslicarbazepine is 10-20 hours in healthy subjects and 13-20 hours in epilepsy patients. Steady-state plasma concentrations are attained after 4 to 5 days of once daily dosing.
Eslicarbazepine acetate is converted to the active metabolite eslicarbazepine which carries out its anticonvulsant activity. The exact mechanism of action is unknown, but it is thought to involve the inhibition of voltage-gated sodium channels. In in vitro electrophysiological studies, eslicarbazepine was shown to inhibit repeated neuronal firing by stabilizing the inactivated state of voltage-gated sodium channels and preventing their return to the activated state. In vitro studies also showed eslicarbazepine inhibiting T-type calcium channels, which likely also has a role in anticonvulsant activity.

LOOKING FOR A SUPPLIER?
