
1. Apimid
2. Apo Flutamide
3. Apo-flutamide
4. Apoflutamide
5. Chimax
6. Cytamid
7. Drogenil
8. Euflex
9. Eulexin
10. Eulexine
11. Fluken
12. Flulem
13. Flumid
14. Fluta 1a Pharma
15. Fluta Cell
16. Fluta Gry
17. Fluta-cell
18. Fluta-gry
19. Flutacell
20. Flutagry
21. Flutamin
22. Flutandrona
23. Flutaplex
24. Flutexin
25. Fugerel
26. Grisetin
27. Niftolid
28. Niftolide
29. Novo Flutamide
30. Novo-flutamide
31. Novoflutamide
32. Oncosal
33. Pms Flutamide
34. Pms-flutamide
35. Prostacur
36. Prostica
37. Prostogenat
38. Sch 13521
39. Sch-13521
40. Sch13521
41. Testotard
1. 13311-84-7
2. Eulexin
3. Niftolide
4. Niftholide
5. Sch 13521
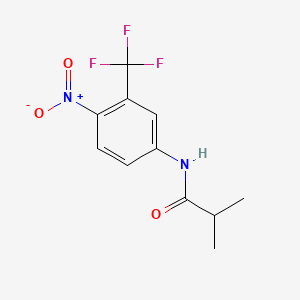
6. 2-methyl-n-[4-nitro-3-(trifluoromethyl)phenyl]propanamide
7. Nfba
8. Niftolid
9. Drogenil
10. Flutamin
11. Cebatrol, Veterinary
12. Flutamida
13. Flutamidum
14. Sch-13521
15. 4'-nitro-3'-trifluoromethylisobutyranilide
16. 2-methyl-n-(4-nitro-3-[trifluoromethyl]phenyl)propanamide
17. N-(4-nitro-3-(trifluoromethyl)phenyl)isobutyramide
18. Sch13521
19. Mfcd00072009
20. Nsc 215876
21. Flutamide (eulexin)
22. Alpha,alpha,alpha-trifluoro-2-methyl-4'-nitro-m-propionotoluidide
23. Chembl806
24. Propanamide, 2-methyl-n-(4-nitro-3-(trifluoromethyl)phenyl)-
25. Propanamide, 2-methyl-n-[4-nitro-3-(trifluoromethyl)phenyl]-
26. Nsc-215876
27. 4-nitro-3-(trifluoromethyl)isobutyranilide
28. Chebi:5132
29. 76w6j0943e
30. Ncgc00015452-09
31. Eulexine
32. Chimax
33. 4'-nitro-3'-trifluoromethylisobutyramilide
34. Cas-13311-84-7
35. Ham's F-12 Medium
36. Flutamidum [inn-latin]
37. Dsstox_cid_11121
38. Dsstox_rid_78899
39. Dsstox_gsid_32004
40. Flutamida [inn-spanish]
41. Flutamide Usp25
42. N-[4-nitro-3-(trifluoromethyl)phenyl]isobutyramide
43. Prostandril
44. Odyne
45. .alpha.,.alpha.,.alpha.-trifluoro-2-methyl-4'-nitro-m-propionotoluidide
46. Smr000058187
47. Eulexin (tn)
48. Ccris 7246
49. M-propionotoluidide,.alpha.,.alpha.-trifluoro-
50. Sr-01000075888
51. 4'-nitro-3'-(trifluoromethyl)isobutyranilide
52. Einecs 236-341-9
53. Brn 2157663
54. .alpha.,.alpha.-trifluoro-2-methyl-4'-nitro-m-propionotoluidide
55. M-propionotoluidide,.alpha.,.alpha.-trifluoro-2-methyl-4'-nitro-
56. Unii-76w6j0943e
57. Flutamide [usan:usp:inn:ban]
58. Flutamide,(s)
59. Prestwick_228
60. Nk-601
61. Spectrum_001210
62. 2-methyl-n-[4-nitro-3-(trifluoromethyl)phenyl]propionamide
63. Cpd000058187
64. Flutamide [inn]
65. Flutamide [jan]
66. Flutamide [mi]
67. Flutamide [usan]
68. Prestwick0_000180
69. Prestwick1_000180
70. Prestwick2_000180
71. Prestwick3_000180
72. Spectrum2_001201
73. Spectrum3_001421
74. Spectrum4_000829
75. Spectrum5_001450
76. Lopac-f-9397
77. F0663
78. Flutamide (pubertal Study)
79. Flutamide [mart.]
80. F 9397
81. Flutamide [usp-rs]
82. Flutamide [who-dd]
83. Schembl3934
84. Lopac0_000557
85. Bspbio_000079
86. Bspbio_003122
87. Kbiogr_001377
88. Kbioss_001690
89. Mls000069634
90. Mls001065596
91. Mls002548892
92. Divk1c_000459
93. Spectrum1500995
94. Spbio_000982
95. Spbio_002000
96. Flutamide (jp17/usp/inn)
97. Bpbio1_000087
98. Gtpl6943
99. Flutamide [ep Impurity]
100. Flutamide [orange Book]
101. Chembl4759307
102. Dtxsid7032004
103. Flutamide [ep Monograph]
104. Schembl12932289
105. Hms501g21
106. Kbio1_000459
107. Kbio2_001690
108. Kbio2_004258
109. Kbio2_006826
110. Kbio3_002342
111. Flutamide [usp Monograph]
112. Ninds_000459
113. Hms1568d21
114. Hms1921o16
115. Hms2090i18
116. Hms2092o14
117. Hms2095d21
118. Hms2230p19
119. Hms3259i03
120. Hms3261p15
121. Hms3373c12
122. Hms3655g22
123. Hms3712d21
124. Pharmakon1600-01500995
125. Amy32524
126. Bcp23006
127. Hy-b0022
128. Zinc3812944
129. Tox21_110154
130. Tox21_202169
131. Tox21_300536
132. Tox21_500557
133. Bdbm50131270
134. Ccg-39105
135. M-propionotoluidide, 2-methyl-4'-nitro-alpha,alpha,alpha-triflouro-
136. M-propionotoluidide, Alpha,alpha,alpha-trifluoro-2-methyl-4'-nitro-
137. Nsc147834
138. Nsc215876
139. Nsc757817
140. S1908
141. Akos001025465
142. Akos025243203
143. M-propionotoluidide, .alpha.,.alpha.,.alpha.-trifluoro-2-methyl-4'-nitro-
144. Tox21_110154_1
145. Ab02835
146. Db00499
147. Ks-5091
148. Lp00557
149. Nc00451
150. Nsc-147834
151. Nsc-757817
152. Sdccgsbi-0050540.p004
153. Idi1_000459
154. Ncgc00015452-01
155. Ncgc00015452-02
156. Ncgc00015452-03
157. Ncgc00015452-04
158. Ncgc00015452-05
159. Ncgc00015452-06
160. Ncgc00015452-07
161. Ncgc00015452-08
162. Ncgc00015452-10
163. Ncgc00015452-11
164. Ncgc00015452-12
165. Ncgc00015452-13
166. Ncgc00015452-14
167. Ncgc00015452-15
168. Ncgc00015452-16
169. Ncgc00015452-19
170. Ncgc00015452-20
171. Ncgc00015452-32
172. Ncgc00091460-01
173. Ncgc00091460-02
174. Ncgc00091460-03
175. Ncgc00091460-04
176. Ncgc00091460-05
177. Ncgc00091460-06
178. Ncgc00091460-07
179. Ncgc00091460-08
180. Ncgc00091460-09
181. Ncgc00254495-01
182. Ncgc00259718-01
183. Ncgc00261242-01
184. Ac-24192
185. Bf166239
186. Sy036411
187. Sbi-0050540.p003
188. Db-042163
189. 3'-trifluoromethyl-4'-nitro-isobutyranilide
190. Ab00052188
191. Eu-0100557
192. Ft-0626493
193. Ft-0668764
194. Sw196536-4
195. 4''-nitro-3''-trifluoromethylisobutyranilide
196. C07653
197. D00586
198. Ab00052188-09
199. Ab00052188_10
200. Ab00052188_11
201. 311f847
202. A806562
203. Q418669
204. Q-201131
205. Sr-01000075888-1
206. Sr-01000075888-6
207. Sr-01000075888-7
208. Sr-01000075888-9
209. Brd-k28307902-001-05-0
210. Flutamide, Certified Reference Material, Tracecert(r)
211. Z56755651
212. A,a,a-trifluoro-2-methyl-4'-nitro- M-propionotoluidide
213. Flutamide, European Pharmacopoeia (ep) Reference Standard
214. 2-methyl-n-[4-nitro-3-(trifluoromethyl)phenyl]propanamide #
215. Flutamide, United States Pharmacopeia (usp) Reference Standard
216. 2-methyl-n-[4-nitro-3-(trifluoromethyl)phenyl]propanamide;flutamide
217. Alpha,alpha,alpha-trifluoro-2-methyl-4''-nitro-m-propionotoluidide
218. M-propionotoluidide, 2-methyl-4'-nitro-.alpha.,.alpha.,.alpha.-trifluoro-
219. Flutamide For System Suitability, European Pharmacopoeia (ep) Reference Standard
220. 37209-54-4
| Molecular Weight | 276.21 g/mol |
|---|---|
| Molecular Formula | C11H11F3N2O3 |
| XLogP3 | 3.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Exact Mass | 276.07217670 g/mol |
| Monoisotopic Mass | 276.07217670 g/mol |
| Topological Polar Surface Area | 74.9 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 352 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Flutamide |
| PubMed Health | Flutamide (By mouth) |
| Drug Classes | Antiandrogen |
| Drug Label | Flutamide capsules contain flutamide, an acetanilid, nonsteroidal, orally active antiandrogen having the chemical name, ,,-trifluoro-2-methyl-4'-nitrom-propionotoluidide.Each capsule contains 125 mg flutamide. The compound is a buff to yellow p... |
| Active Ingredient | Flutamide |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 125mg |
| Market Status | Prescription |
| Company | Actavis Labs Fl; Par Pharm; Ivax Sub Teva Pharms; Mylan |
| 2 of 2 | |
|---|---|
| Drug Name | Flutamide |
| PubMed Health | Flutamide (By mouth) |
| Drug Classes | Antiandrogen |
| Drug Label | Flutamide capsules contain flutamide, an acetanilid, nonsteroidal, orally active antiandrogen having the chemical name, ,,-trifluoro-2-methyl-4'-nitrom-propionotoluidide.Each capsule contains 125 mg flutamide. The compound is a buff to yellow p... |
| Active Ingredient | Flutamide |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 125mg |
| Market Status | Prescription |
| Company | Actavis Labs Fl; Par Pharm; Ivax Sub Teva Pharms; Mylan |
For the management of locally confined Stage B2-C and Stage D2 metastatic carcinoma of the prostate
FDA Label
Flutamide is a nonsteroidal antiandrogen. In animal studies, flutamide demonstrates potent antiandrogenic effects. It exerts its antiandrogenic action by inhibiting androgen uptake and/or by inhibiting nuclear binding of androgen in target tissues or both. Prostatic carcinoma is known to be androgen-sensitive and responds to treatment that counteracts the effect of androgen and/or removes the source of androgen, e.g. castration. Elevations of plasma testosterone and estradiol levels have been noted following flutamide administration.
Antineoplastic Agents, Hormonal
Antineoplastic agents that are used to treat hormone-sensitive tumors. Hormone-sensitive tumors may be hormone-dependent, hormone-responsive, or both. A hormone-dependent tumor regresses on removal of the hormonal stimulus, by surgery or pharmacological block. Hormone-responsive tumors may regress when pharmacologic amounts of hormones are administered regardless of whether previous signs of hormone sensitivity were observed. The major hormone-responsive cancers include carcinomas of the breast, prostate, and endometrium; lymphomas; and certain leukemias. (From AMA Drug Evaluations Annual 1994, p2079) (See all compounds classified as Antineoplastic Agents, Hormonal.)
Androgen Antagonists
Compounds which inhibit or antagonize the biosynthesis or actions of androgens. (See all compounds classified as Androgen Antagonists.)
L02BB01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L02 - Endocrine therapy
L02B - Hormone antagonists and related agents
L02BB - Anti-androgens
L02BB01 - Flutamide
Absorption
Rapidly and completely absorbed.
Route of Elimination
Flutamide and its metabolites are excreted mainly in the urine with only 4.2% of a single dose excreted in the feces over 72 hours.
Flutamide is rapidly and extensively metabolized, with flutamide comprising only 2.5% of plasma radioactivity 1 hour after administration.
Flutamide has known human metabolites that include 2-hydroxy-flutamide.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The plasma half-life for the alpha-hydroxylated metabolite of flutamide (an active metabolite) is approximately 6 hours.
Flutamide is a nonsteroidal antiandrogen that blocks the action of both endogenous and exogenous testosterone by binding to the androgen receptor. In addition Flutamide is a potent inhibitor of testosterone-stimulated prostatic DNA synthesis. Moreover, it is capable of inhibiting prostatic nuclear uptake of androgen.