


1. Abt-493
1. Abt-493
2. Glecaprevir [usan]
3. 1365970-03-1
4. A-1282576.0
5. K6buu8j72p
6. A-1282576
7. A-12825760
8. 1365970-03-1 (free)
9. Abt 493
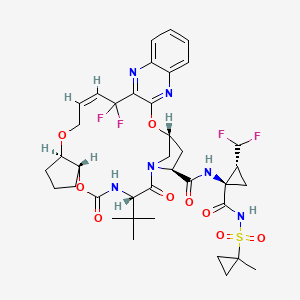
10. (3ar,7s,10s,12r,21e,24ar)-7-tert-butyl-n-[(1r,2r)-2-(difluoromethyl)-1-{[(1-methylcyclopropyl)sulfonyl]carbamoyl}cyclopropyl]-20,20-difluoro-5,8-dioxo-2,3,3a,5,6,7,8,11,12,20,23,24a-dodecahydro-1h,10h-9,12-methanocyclopenta[18,19][1,10,17,3,6]trioxadiazacyclononadecino[11,12-b]quinoxaline-10-carboxamide
11. (3ar,7s,10s,12r,21e,24ar)-7-tert-butyl-n-{(1r,2r)-2(difluoromethyl)-1-[(1-methylcyclopropane-1-sulfonyl)carbamoyl]cyclopropyl}-20,20-difluoro5,8-dioxo-2,3,3a,5,6,7,8,11,12,20,23,24a-dodecahydro-1h,10h-9,12methanocyclopenta[18,19][1,10,17,3,6]trioxadiazacyclononadecino[11,12-b]quinoxaline-10carboxamide
12. Unii-k6buu8j72p
13. Maviret
14. (3ar,7s,10s,12r,21e,24ar)-7-tert-butyl-n-[(1r,2r)-2-(difluoromethyl)-1-{[(1-methylcyclopropyl)sulfonyl]carbamoyl}cyclop Ropyl]-20,20-difluoro-5,8-dioxo-2,3,3a,5,6,7,8,11,12,20,23,24a-dodecahydro-1h,10h-9,12-methanocyclopenta[18,19][1,10,17, 3,6]trioxadiazacyclononadecino[11,12-b]quinoxaline-10-carboxamide
15. O31
16. Glecaprevir [mi]
17. Abt-493(glecaprevir)
18. Glecaprevir [inn]
19. Glecaprevir (usan/inn)
20. Glecaprevir [who-dd]
21. Schembl883097
22. Abt493
23. Chembl3545363
24. Gtpl11267
25. Glecaprevir [orange Book]
26. Dtxsid901027945
27. (1r,14e,18r,22r,26s,29s)-26-tert-butyl-n-[(1r,2r)-2-(difluoromethyl)-1-[(1-methylcyclopropyl)sulfonylcarbamoyl]cyclopropyl]-13,13-difluoro-24,27-dioxo-2,17,23-trioxa-4,11,25,28-tetrazapentacyclo[26.2.1.03,12.05,10.018,22]hentriaconta-3,5,7,9,11,14-hexaene-29-carboxamide
28. Amy38157
29. Ex-a1940
30. Mavyret Component Glecaprevir
31. Bdbm50573891
32. S5720
33. Cs-8098
34. Db13879
35. Ac-33419
36. Hy-17634
37. J3.646.120i
38. D10814
39. 3,6]trioxadiazacyclononadecino[11,12-b]quinoxaline-10-carboxamide
40. (3ar,7s,10s,12r,21e,24ar)-7-tert-butyl-n-((1r,2r)-2-(difluoromethyl)-1-((1-methylcyclopropane-1-sulfonyl)carbamoyl)cyclopropyl)-20,20-difluoro-5,8-dioxo-2,3,3a,5,6,7,8,11,12,20,23,24a-dodecahydro-1h,10h-9,12-methanocyclopenta(18,19)(1,10,17,3,6)trioxadiazacyclononadecino(11,12-b)quinoxaline-10-carboxamide
41. (3ar,7s,10s,12r,21e,24ar)-7-tert-butyl-n-[(1r,2r)-2-(difluoromethyl)-1-{[(1-methylcyclopropyl)sulfonyl]carbamoyl}cyclop
42. (3ar,7s,10s,12r,21e,24ar)-7-tert-butyl-n-{(1r,2r)-2- (difluoromethyl)-1-[(1-methylcyclopropane-1-sulfonyl)carbamoyl]cyclopropyl}-20,20-difluoro- 5,8-dioxo-2,3,3a,5,6,7,8,11,12,20,23,24a-dodecahydro-1h,10h-9,12- Methanocyclopenta[18,19][1,10,17,3,6]trioxadiazacyclononadecino[11,12-b]quinoxaline-10- Carboxamide Hydrate
43. Cyclopropanecarboxamide, N-((((1r,2r)-2-((4,4-difluoro-4-(3-hydroxy-2-quinoxalinyl)-2-buten-1-yl)oxy)cyclopentyl)oxy)carbonyl)-3-methyl-l-valyl-(4r)-4-hydroxy-l-prolyl-1-amino-2-(difluoromethyl)-n-((1-methylcyclopropyl)sulfonyl)-, Cyclic (1->2)-ether, (1r,2r)-
44. Ropyl]-20,20-difluoro-5,8-dioxo-2,3,3a,5,6,7,8,11,12,20,23,24a-dodecahydro-1h,10h-9,12-methanocyclopenta[18,19][1,10,17,
| Molecular Weight | 838.9 g/mol |
|---|---|
| Molecular Formula | C38H46F4N6O9S |
| XLogP3 | 4.6 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 7 |
| Exact Mass | 838.29831089 g/mol |
| Monoisotopic Mass | 838.29831089 g/mol |
| Topological Polar Surface Area | 204 Ų |
| Heavy Atom Count | 58 |
| Formal Charge | 0 |
| Complexity | 1760 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 7 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Indicated for the treatment of adult patients with chronic hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5 or 6 infection without cirrhosis or with compensated cirrhosis (Child-Pugh A). MAVYRET is also indicated for the treatment of adult patients with HCV genotype 1 infection, who previously have been treated with a regimen containing an HCV NS5A inhibitor or an NS3/4A protease inhibitor (PI), but not both.
FDA Label
Maviret is indicated for the treatment of chronic hepatitis C virus (HCV) infection in adults and children aged 3 years and older.
Maviret coated granules is indicated for the treatment of chronic hepatitis C virus (HCV) infection in children 3 years and older.
In a biochemical assay studying clinical isolates of HCV genotypes 1a, 1b, 2a, 2b, 3a, 4a, 5a, and 6a, glecaprevir displayed IC50 values ranging from 3.5 to 11.3 nM that resulted in inhibition of the proteolytic activity of recombinant NS3/4A enzymes. In HCV replicon assays, glecaprevir had median EC50 values of 0.08-4.6 nM against laboratory and clinical isolates from subtypes 1a, 1b, 2a, 2b, 3a, 4a, 4d, 5a, and 6a. In a QT study, glecaprevir is not shown to prolong the QTc interval.
J05AP57
Absorption
In healthy subjects, the time it takes to reach the peak plasma concentration (Tmax) is approximately 5 hours. The mean peak plasma concentration (Cmax) is 597ng/mL in non-cirrhotic HCV-infected subjects. Relative to fasting conditions, the consumption of meals increases the absorption of glecaprevir by 83-163%.
Route of Elimination
The predominant route of elimination of the drug is biliary-fecal, where 92.1% of administered drug is excreted in feces and 0.7% of the drug is excreted in the urine.
Glecaprevir undergoes limited secondary metabolism in vitro, predominantly by CYP3A.
The elimination half life (t1/2) is approximately 6 hours.
Glecaprevir is an inhibitor of the HCV NS3/4A protease, which is a viral enzyme necessary for the proteolytic cleavage of the HCV encoded polyprotein into mature forms of the NS3, NS4A, NS4B, NS5A, and NS5B proteins. These multifunctional proteins, including NS3, are essential for viral replication. The N-terminal of NS3 protein confers serine protease activity, whileThe C-terminus of NS3 encodes a DExH/D-box RNA helicase which hydyolyzes NTP as an energy source to unwind double-stranded RNA in a 3 to 5 direction during replication of viral genomic RNA. NS4A is a cofactor for NS3 that directs the localization of NS3 and modulates its enzymatic activities. Glecaprevir disrupts the intracellular processes of the viral life cycle through inhibiting the NS3/4A protease activity of cleaving downstream junctions of HCV polypeptide and proteolytic processing of mature structural proteins.
