
1. Contrast Media 2-5410-3
2. Iodixanol-320
3. Visipaque
4. Visipaque Unique Softpac
1. 92339-11-2
2. Visipaque
3. Indixanol
4. Iodixanolum
5. 2-5410-3a
6. Hw8w27htxx
7. Nsc-760069
8. 5,5'-((2-hydroxytrimethylene)bis(acetylimino))bis(n,n'-bis(2,3-dihydroxypropyl)-2,4,6-triiodoisophthalamide)
9. Chebi:31705
10. 5-[acetyl-[3-[n-acetyl-3,5-bis(2,3-dihydroxypropylcarbamoyl)-2,4,6-triiodoanilino]-2-hydroxypropyl]amino]-1-n,3-n-bis(2,3-dihydroxypropyl)-2,4,6-triiodobenzene-1,3-dicarboxamide
11. 2-541o-3a
12. Dsstox_cid_25523
13. Dsstox_rid_80929
14. Dsstox_gsid_45523
15. 1,3-benzenedicarboxamide, 5,5'-((2-hydroxy-1,3-propanediyl)bis(acetylimino))bis(n,n'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-
16. 5-[acetyl-[3-[n-acetyl-3,5-bis[[(2s)-2,3-dihydroxypropyl]carbamoyl]-2,4,6-triiodoanilino]-2-hydroxypropyl]amino]-1-n,3-n-bis[(2s)-2,3-dihydroxypropyl]-2,4,6-triiodobenzene-1,3-dicarboxamide
17. Optiprep; Visipaque; Visipaque 270;2-5410-3a
18. Visipaque 270
19. Visipaque 320
20. Iodixanolum [latin]
21. Optiprep
22. 1,3-benzenedicarboxamide, 5,5'-[(2-hydroxy-1,3-propanediyl)bis(acetylimino)]bis[n,n'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-
23. 5,5'-((2-hydroxypropane-1,3-diyl)bis(acetylazanediyl))bis(n1,n3-bis(2,3-dihydroxypropyl)-2,4,6-triiodoisophthalamide)
24. Ccris 7567
25. Sr-01000872678
26. Unii-hw8w27htxx
27. Hsdb 8076
28. Iodixanol [usan:usp:inn:ban]
29. Visipaque (tn)
30. Ncgc00016958-01
31. Cas-92339-11-2
32. Visipaque;iodixanolum
33. Iodixanol [inn]
34. Iodixanol [jan]
35. Iodixanol [mi]
36. Iodixanol [usan]
37. Prestwick0_000848
38. Prestwick1_000848
39. Prestwick2_000848
40. Prestwick3_000848
41. Iodixanol [vandf]
42. Iodixanol [mart.]
43. Ec 618-837-0
44. Iodixanol [usp-rs]
45. Iodixanol [who-dd]
46. Iodixanol (jan/usp/inn)
47. Schembl21546
48. Bspbio_000835
49. Spbio_002756
50. Bpbio1_000919
51. Iodixanol [orange Book]
52. Chembl1200507
53. Dtxsid2045523
54. Iodixanol [ep Monograph]
55. Iodixanol [usp Impurity]
56. Iodixanol [usp Monograph]
57. Hms1570j17
58. Hms2097j17
59. Hms3714j17
60. Pharmakon1600-01503835
61. Bcp11111
62. Hy-b1426
63. Tox21_110711
64. Nsc760069
65. S5218
66. Akos015895607
67. Tox21_110711_1
68. Ac-7610
69. Ccg-213207
70. Db01249
71. Nsc 760069
72. Ncgc00179408-01
73. Ncgc00179408-03
74. 5-(acetyl-(3-(acetyl-(3,5-bis(2,3-dihydroxypropylcarbamoyl)-2,4,6-triiodophenyl)amino)-2- Hydroxypropyl)amino)-n,n'-bis(2,3-dihydroxypropyl)-2,4,6-triiodobenzene-1,3-dicarboxamide; 5,5'-((2-hydroxytrimethylene)bis(acetylimino))bis(n,n'-bis(2,3-dihydroxypropyl)-2,4,6- Triiodoisophthalamide);
75. Ab00513936
76. Cs-0013137
77. Ft-0627254
78. Ft-0645028
79. D01474
80. Ab00513936_02
81. 339i112
82. A844203
83. Q2419291
84. Sr-01000872678-1
85. Sr-01000872678-2
86. Brd-a08660406-001-01-8
87. Optiprep(tm) Density Gradient Medium, Used For Cell And Subcellular Organelle Isolation
88. 5,5'-(2-hydroxypropane-1,3-diyl)bis(acetylazanediyl)bis(n1,n3-bis(2,3-dihydroxypropyl)-2,4,6-triiodoisophthalamide)
89. 5,5'-[(2-hydroxypropane-1,3-diyl)bis(acetylimino)]bis[n,n'-bis(2,3-dihydroxypropyl)-2,4,6-triiodobenzene-1,3-dicarboxamide]
90. 5,5-[(2-hydroxy-1,3-propanediyl)bis(acetylimino)]bis[n,n-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
91. 5-[[3-[[3,5-bis[2,3-bis(oxidanyl)propylcarbamoyl]-2,4,6-tris(iodanyl)phenyl]-ethanoyl-amino]-2-oxidanyl-propyl]-ethanoyl-amino]-n1,n3-bis[2,3-bis(oxidanyl)propyl]-2,4,6-tris(iodanyl)benzene-1,3-dicarboxamide
92. 5-[acetyl-[3-[n-acetyl-3,5-bis[(2,3-dihydroxypropylamino)-oxomethyl]-2,4,6-triiodoanilino]-2-hydroxypropyl]amino]-n1,n3-bis(2,3-dihydroxypropyl)-2,4,6-triiodobenzene-1,3-dicarboxamide
93. 5-{n-[3-(n-{3,5-bis[(2,3-dihydroxypropyl)carbamoyl]-2,4,6-triiodophenyl}acetamido)-2-hydroxypropyl]acetamido}-1-n,3-n-bis(2,3-dihydroxypropyl)-2,4,6-triiodobenzene-1,3-dicarboxamide
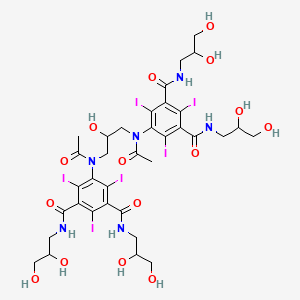
| Molecular Weight | 1550.2 g/mol |
|---|---|
| Molecular Formula | C35H44I6N6O15 |
| XLogP3 | -3.4 |
| Hydrogen Bond Donor Count | 13 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 22 |
| Exact Mass | 1549.7133 g/mol |
| Monoisotopic Mass | 1549.7133 g/mol |
| Topological Polar Surface Area | 339 Ų |
| Heavy Atom Count | 62 |
| Formal Charge | 0 |
| Complexity | 1330 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 4 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Visipaque 270 |
| Active Ingredient | Iodixanol |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 55% |
| Market Status | Prescription |
| Company | Ge Healthcare |
| 2 of 4 | |
|---|---|
| Drug Name | Visipaque 320 |
| Active Ingredient | Iodixanol |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 65.2% |
| Market Status | Prescription |
| Company | Ge Healthcare |
| 3 of 4 | |
|---|---|
| Drug Name | Visipaque 270 |
| Active Ingredient | Iodixanol |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 55% |
| Market Status | Prescription |
| Company | Ge Healthcare |
| 4 of 4 | |
|---|---|
| Drug Name | Visipaque 320 |
| Active Ingredient | Iodixanol |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 65.2% |
| Market Status | Prescription |
| Company | Ge Healthcare |
Iodixanol is indicated for contrast enhanced computerized tomography imaging of the head and body, excretory urography, and peripheral venography. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
Iodixanol is indicated for angiocardiography (left ventriculography and selective coronary arteriography), peripheral arteriography, visceral arteriography, and cerebral arteriography. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
Iodixanol is indicated for intra-arterial digital subtraction angiography. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
Iodixanol is indicated for contrast enhanced computerized tomography imaging of the head and body, and excretory urography. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
For more Therapeutic Uses (Complete) data for Iodixanol (6 total), please visit the HSDB record page.
The following selected adverse events were reported in ?0.5% of the 1244 patients in controlled clinical trials who received iodixanol injection: General Disorders: back pain, fatigue, malaise; Cardiovascular Disorders: arrhythmias, cardiac failure, conduction abnormalities, hypotension, myocardial infarction; Nervous System: cerebral vascular disorder, convulsions, hypoesthesia, stupor, confusion; Gastrointestinal System Disorders: dyspepsia; Hypersensitivity Disorders: pharyngeal edema; Respiratory System Disorders: asthma, bronchitis, dyspnea, pulmonary edema, rhinitis; Renal System Disorders: abnormal renal function, acute renal failure, hematuria; Peripheral Vascular Disorders: flushing, peripheral ischemia; Skin and Appendage Disorders: hematoma, increased sweating; Special Senses, Other Disorders: tinnitus; Vision Disorders: abnormal vision.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
Additional adverse events reported in other clinical studies and in foreign postmarketing surveillance and foreign clinical trials with the use of iodixanol injection are: anaphylactic reactions, anaphylactoid reactions, hypoglycemia, amnesia, cardiac arrest, hypertension, dyskinesia, hemorrhage not otherwise specified, polymyalgia rheumatica, pulmonary embolism, respiratory depression, and cortical blindness.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
... Selected commonly reported adverse events in pediatrics include: vomiting, nausea, fever, rash, and pruritus. Less frequently reported events are apnea, disseminated intravascular coagulation, atrioventricular block and bundle branch block, arrhythmia, cardiac failure, renal failure and taste perversion.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
In patients with normal blood-brain barriers and renal failure, iodinated contrast agents have been associated with blood-brain barrier disruption and accumulation of contrast in the brain.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
For more Drug Warnings (Complete) data for Iodixanol (28 total), please visit the HSDB record page.
Iodixanol is a contrast agent during coronary angiography.
FDA Label
Iodixanol is a contrast agent commonly used during coronary angiography, particularly in individuals with renal dysfunction, as it is believed to be less toxic to the kidneys than most other intravascular contrast agents. It is an imaging contrast agent with the same osmolality as blood (290mOsm/kg H20).
Contrast Media
Substances used to allow enhanced visualization of tissues. (See all compounds classified as Contrast Media.)
V08AB09
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
V - Various
V08 - Contrast media
V08A - X-ray contrast media, iodinated
V08AB - Watersoluble, nephrotropic, low osmolar x-ray contrast media
V08AB09 - Iodixanol
Route of Elimination
In adults, approximately 97% of the injected dose of iodixanol is excreted unchanged in urine within 24 hours, with less than 2% excreted in feces within five days post-injection.
Volume of Distribution
0.26 L/kg
Absorption and excretion of iodixanol 320 mg I/ml were investigated in rats after intragastric administration of 2.5 g I/kg b.w. Animals were observed for up to 96 hours after treatment, and blood, urine and feces taken at several time-points throughout the experiment. Concentrations of iodixanol in serum and urine were measured by means of reversed-phase high-performance liquid chromatography. Fecal concentrations of iodixanol, based on iodine measurements, were determined by X-ray fluorescence spectrometry. Serial radiographs were obtained and histopathological examination was performed on selected tissues. The results indicate that less than 1% of the intragastric dose of iodixanol is absorbed from the intestine into the blood stream. No adverse clinical signs were observed, and there were no treatment-related histomorphological findings.
PMID:8610524 Dunkel JA et al; Acta Radiol Suppl 399: 253-7 (1995)
In 40 healthy, young male volunteers receiving a single intravenous administration of iodixanol injection in doses of 0.3 to 1.2 gI/kg body weight, ... renal clearance was 110 mL/min (+/-14), equivalent to glomerular filtration (108 mL/min). These values were independent of the dose administered.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
... In a study of 16 adult patients who were scheduled for renal transplant, the elimination of iodixanol 320 mgI/mL was studied. The patients' baseline mean creatinine levels were 6.3 mg/dL (+/-1.5) and mean creatinine clearances were 13.61 mL/min (+/- 4.67). ... In these patients, levels of iodixanol were detected 5 days after dosing. Contrast enhancement time in kidneys increased from 6 hours to at least 24 hours. ...
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
Plasma and urine levels suggest that body clearance of iodixanol is primarily due to renal clearance. In adults, approximately 97% of the injected dose of iodixanol is excreted unchanged in urine within 24 hours, with less than 2% excreted in feces within five days post-injection.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
For more Absorption, Distribution and Excretion (Complete) data for Iodixanol (14 total), please visit the HSDB record page.
Excreted unchanged
Iodixanol metabolites have not been demonstrated.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
2.1 hours. In patients with significantly impaired renal function (mean creatinine clearance rate, 9.91 [± 3.58] mL per minute), the plasma half-life is increased to 23 hours.
Forty pediatric patients < or = 12 years old, with renal function that is normal for their age, received multiple intra-arterial administrations of iodixanol injection in doses of 0.32 to 3.2 gI/kg body weight. The elimination half-lives for these patients are ...: 0.185 hr -1 (newborn to 2 months old), 0.256 hr -1 (2 to <6 months old), 0.299 hr -1 (6 months to<1 year), 0.322 hr -1 (1 to<2 years), and 0.307 hr -1 (2 to < or = 12 years old). The adult mean terminal elimination rate constant is 0.336 hr-1. ...
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
In 40 healthy, young male volunteers receiving a single intravenous administration of iodixanol injection in doses of 0.3 to 1.2 gI/kg body weight, the elimination half-life was 2.1 hr (+/- 0.1) ...
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
... Iodixanol was rapidly excreted, mainly via the kidneys, with a plasma half-life in rats and monkeys of 25 and 76 mins, respectively. ...
PMID:8610532 Heglund IF et al; Acta Radiol Suppl 399: 69-82 (1995)
... In a study of 16 adult patients who were scheduled for renal transplant, the elimination of iodixanol 320 mgI/mL was studied. ... In these patients, the plasma half-life was increased to 23 hours (normal t1/2 = 2 hours). ...
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e
Organic iodine compounds attenuate x-rays as they pass through the body, thereby allowing the body structures containing iodine to be delineated in contrast to those structures that do not contain iodine. The degree of opacity produced by these compounds is directly proportional to the total amount (concentration and volume) of the iodinated contrast agent in the path of the x-rays. After intravascular administration, iodixanol makes opaque those internal structures in its path of flow, allowing their visualization until significant hemodilution and elimination occur.
Intravascular injection of iodixanol opacifies those vessels in the path of flow of the contrast agent, permitting radiographic visualization of the internal structures until significant dilution and elimination occurs.
US Natl Inst Health; DailyMed. Current Medication Information for VISIPAQUE (iodixanol) injection, solution (May 2009). Available from, as of July 11, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2d964c13-88ad-43be-88d4-a3a7ea4d5b5e