

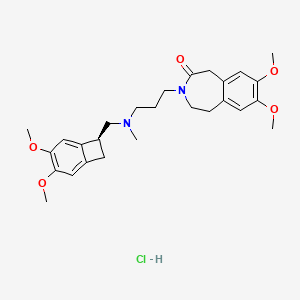

1. 7,8-dimethoxy-3-(3-(((4,5-dimethoxybenzocyclobutan-1-yl)methyl)methylamino)propyl)-1,3,4,5-tetrahydro-2h-benzazepin-2-one
2. Corlanor
3. Ivabradine
4. S 16257
5. S 16257 2
6. S 16257-2
7. S 162572
8. S 16260 2
9. S 16260-2
10. S 162602
11. S-16257
12. S-16257-2
13. S-16260-2
14. S16257
15. S162572
16. S162602
1. 148849-67-6
2. Ivabradine Hcl
3. Procoralan
4. Corlentor
5. Corlanor
6. Ivabradine (hydrochloride)
7. Tp19837bzk
8. (s)-3-(3-(((3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl)methyl)(methyl)amino)propyl)-7,8-dimethoxy-1,3,4,5-tetrahydro-2h-benzo[d]azepin-2-one Hydrochloride
9. Ivabradine Hydrochloride [usan]
10. Chebi:85969
11. Amg 998
12. Amg-998
13. S-16257-2
14. 2h-3-benzazepin-2-one, 3-[3-[[[(7s)-3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl]methyl]methylamino]propyl]-1,3,4,5-tetrahydro-7,8-dimethoxy-, Hydrochloride (1:1)
15. Ncgc00181343-01
16. 148849-67-6 (hcl)
17. (s)-3-(3-(((3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl)methyl)(methyl)amino)propyl)-7,8-dimethoxy-4,5-dihydro-1h-benzo[d]azepin-2(3h)-one Hydrochloride
18. 3-[3-[[[(7s)-3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl]methyl]methylamino]propyl]-1,3,4,5-tetrahydro-7,8-dimethoxy-2h-3-benzazepin-2-one Hydrochloride
19. Ivabradinehydrochloride
20. Ivabradine Hcl (procoralan)
21. S16257
22. Unii-tp19837bzk
23. Corlentor (tn)
24. Corlanor (tn)
25. 2h-3-benzazepin-2-one, 3-(3-((((7s)-3,4-dimethoxybicyclo(4.2.0)octa-1,3,5-trien-7-yl)methyl)methylamino)propyl)-1,3,4,5-tetrahydro-7,8-dimethoxy-, Hydrochloride (1:1)
26. 3-(3-((((7s)-3,4-dimethoxybicyclo(4.2.0)octa-1,3,5-trien-7-yl)methyl)methylamino)propyl)-1,3,4,5-tetrahydro-7,8-dimethoxy-2h-3-benzazepin-2-one Hydrochloride
27. Coralan (tn)
28. Ivabrandine Hydrochloride
29. Ivabradine Monohydrochloride
30. Dsstox_cid_26913
31. Dsstox_rid_82011
32. Dsstox_gsid_46913
33. Schembl23470
34. Mls006010110
35. Ivabradine Hydrochloride- Bio-x
36. Chembl2145077
37. Dtxsid2046913
38. Hy-b0162a
39. 3-[3-[[(7s)-3,4-dimethoxy-7-bicyclo[4.2.0]octa-1,3,5-trienyl]methyl-methylamino]propyl]-7,8-dimethoxy-2,5-dihydro-1h-3-benzazepin-4-one;hydrochloride
40. Bcp05218
41. Tox21_112802
42. Ac-543
43. Ivabradine Hydrochloride (jan/usan)
44. Ivabradine Hydrochloride [mi]
45. Mfcd00929899
46. S2086
47. Ivabradine Hydrochloride [jan]
48. Akos015849582
49. Akos016002249
50. Bcp9000798
51. Ccg-269739
52. Cs-1994
53. Gs-3584
54. Ivabradine Hydrochloride [who-dd]
55. (7,8-dimethoxy 3-(3-(((1s)-(4,5-dimethoxybenzocyclobutan-1-yl)methyl)methylamino)propyl)-1,3,4,5-tetrahydro-2h-benzazepin-2-one Hydrochloride
56. (s)-3-(3-(((4,5-dimethoxy-1,2-dihydrocyclobutabenzen-1-yl)methyl)(methyl)amino)propyl)-7,8-dimethoxy-4,5-dihydro-1h-benzo[d]azepin-2(3h)-one Hydrochloride
57. Bi164596
58. Smr004701246
59. Ivabradine Hydrochloride [ema Epar]
60. Ivabradine Hydrochloride, >=98% (hplc)
61. Am20090729
62. B1360
63. Cas-148849-67-6
64. I0847
65. Ivabradine Hydrochloride [orange Book]
66. D08095
67. 974i008
68. J-008523
69. J-521616
70. Q27158823
71. (+)-s16257
72. 2h-3-benzazepin-2-one, 3-(3-((((7s)-3,4-dimethoxybicyclo(4.2.0)octa-1,3,5-trien-7-yl)methyl)methylamino)propyl)-1,3,4,5-tetrahydro-7,8-dimethoxy-, Monohydrochloride
73. 2h-3-benzazepin-2-one, 3-(3-(((3,4-dimethoxybicyclo(4.2.0)octa-1,3,5-trien-7-yl)methyl)methylamino)propyl)-1,3,4,5-tetrahydro-7,8-dimethoxy-, Monohydrochloride, (s)-
74. 3-[3-({[(7s)-3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl]methyl}(methyl)amino)propyl]-7,8-dimethoxy-2,3,4,5-tetrahydro-1h-3-benzazepin-2-one Hydrochloride
75. 3-[3-[[(8s)-3,4-dimethoxy-8-bicyclo[4.2.0]octa-1,3,5-trienyl]methyl-methylamino]propyl]-7,8-dimethoxy-2,5-dihydro-1h-3-benzazepin-4-one Hydrochloride
76. 3-[3-[[(8s)-3,4-dimethoxy-8-bicyclo[4.2.0]octa-1,3,5-trienyl]methyl-methylamino]propyl]-7,8-dimethoxy-2,5-dihydro-1h-3-benzazepin-4-one Hydrochloride3-[3-[[(8s)-3,4-dimethoxy-8-bicyclo[4.2.0]octa-1,3,
77. 3-{3-[{[(7s)-3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl]-methyl}(methyl)amino]propyl}-7,8-dimethoxy-1,3,4,5-tetrahydro-2h-3-benzazepin-2-one Hydrochloride
78. 3-{3-[{[(7s)-3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl]methyl}(methyl)amino]propyl}-7,8-dimethoxy-1,3,4,5-tetrahydro-2h-3-benzazepin-2-one--hydrogen Chloride (1/1)
79. N-{[(7s)-3,4-dimethoxybicyclo[4.2.0]octa-1,3,5-trien-7-yl]methyl}-3-(7,8-dimethoxy-2-oxo-1,2,4,5-tetrahydro-3h-3-benzazepin-3-yl)-n-methylpropan-1-aminium Chloride
| Molecular Weight | 505.0 g/mol |
|---|---|
| Molecular Formula | C27H37ClN2O5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 10 |
| Exact Mass | 504.2391000 g/mol |
| Monoisotopic Mass | 504.2391000 g/mol |
| Topological Polar Surface Area | 60.5 Ų |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 663 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Symptomatic treatment of chronic stable angina pectoris Ivabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate 70 bpm. Ivabradine is indicated:
- in adults unable to tolerate or with a contra-indication to the use of beta-blockers
or
- in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose. Treatment of chronic heart failure Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated.
* Symptomatic treatment of chronic stable angina pectoris:
Ivabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate 70 bpm. Ivabradine is indicated:
- in adults unable to tolerate or with a contraindication to the use of beta-blockers
- or in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose.
* Treatment of chronic heart failure:
Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated.
Symptomatic treatment of chronic stable angina pectoris
Ivabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate 70 bpm. Ivabradine is indicated:
- in adults unable to tolerate or with a contraindication to the use of beta-blockers
- or in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose.
Treatment of chronic heart failure
Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated.
* Symptomatic treatment of chronic stable angina pectoris:
Ivabradine is indicated for the symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate 70 bpm.
Ivabradine is indicated:
- in adults unable to tolerate or with a contra-indication to the use of beta-blockers
- or in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose.
* Treatment of chronic heart failure:
Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated. (see section 5. 1)
Symptomatic treatment of chronic stable angina pectoris in coronary artery disease adults with normal sinus rhythm and heart rate 70 bpm. Ivabradine is indicated: - in adults unable to tolerate or with a contra-indication to the use of beta-blockers - or in combination with beta-blockers in patients inadequately controlled with an optimal beta-blocker dose. Treatment of chronic heart failure Ivabradine is indicated in chronic heart failure NYHA II to IV class with systolic dysfunction, in patients in sinus rhythm and whose heart rate is 75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated.
Treatment of angina pectoris, Treatment of chronic heart failure, Treatment of coronary artery disease
Treatment of angina pectoris, Treatment of chronic heart failure, Treatment of coronary artery disease
Cardiovascular Agents
Agents that affect the rate or intensity of cardiac contraction, blood vessel diameter, or blood volume. (See all compounds classified as Cardiovascular Agents.)
C01EB17
C01EB17
C01EB17
C01EB17
C01EB17
