


1. Ag-120
2. Ag120
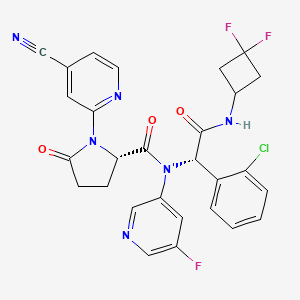
3. N-((1s)-1-(2-chlorophenyl)-2-((3,3-difluorocyclobutyl)amino)-2-oxoethyl)-1-(4-cyano-2-pyridinyl)-n-(5-fluoro-3-pyridinyl)-5-oxo-l-prolinamide
4. Tibsovo
1. 1448347-49-6
2. Tibsovo
3. Ag-120
4. Ag120
5. Ivosidenib [inn]
6. Ivosidenib [usan]
7. Ivosidenib [who-dd]
8. Q2pcn8mam6
9. (2s)-n-[(1s)-1-(2-chlorophenyl)-2-[(3,3-difluorocyclobutyl)amino]-2-oxoethyl]-1-(4-cyanopyridin-2-yl)-n-(5-fluoropyridin-3-yl)-5-oxopyrrolidine-2-carboxamide
10. (s)-n-((s)-1-(2-chlorophenyl)-2-((3,3-difluorocyclobutyl)amino)-2-oxoethyl)-1-(4-cyanopyridin-2-yl)-n-(5-fluoropyridin-3-yl)-5-oxopyrrolidine-2-carboxamide
11. Glycinamide, 1-(4-cyano-2-pyridinyl)-5-oxo-l-prolyl-2-(2-chlorophenyl)-n-(3,3-difluorocyclobutyl)-n2-(5-fluoro-3-pyridinyl)-, (2s)-
12. Rg120
13. Ivosidenibum
14. (2s)-n-((1s)-1-(2-chlorophenyl)-2-((3,3-difluorocyclobutyl)amino)-2-oxoethyl)-1-(4-cyanopyridin-2-yl)-n-(5-fluoropyridin-3-yl)-5-oxopyrrolidine-2-carboxamide
15. (2s)-n-{(1s)-1-(2-chlorophenyl)-2-[(3,3-difluorocyclobutyl)amino]-2-oxoethyl}-1-(4-cyanopyridin-2-yl)-n-(5-fluoropyridin-3-yl)-5-oxopyrrolidine-2-carboxamide
16. Tibsovo (tn)
17. Ivosidenib [mi]
18. Ivosidenib (usan/inn)
19. Ivosidenib [usan:inn]
20. Unii-q2pcn8mam6
21. Gtpl9217
22. Chembl3989958
23. Ivosidenib [orange Book]
24. Schembl15122512
25. Ex-a992
26. Chebi:145430
27. Bdbm363689
28. Dtxsid801027928
29. Amy38924
30. Us9850277, Compound 176
31. Mfcd29036964
32. Nsc789102
33. S8206
34. Zinc205136523
35. Ccg-270141
36. Cs-5122
37. Db14568
38. Nsc-789102
39. Ncgc00476170-04
40. Ncgc00476170-06
41. (s)-n-((s)-1-(2-chlorophenyl)-2-(3,3-difluorocyclobutylamino)-2-oxoethyl)-1-(4-cyanopyridin-2-yl)-n-
42. (s)-n-((s)-1-(2-chlorophenyl)-2-(3,3-difluorocyclobutylamino)-2-oxoethyl)-1-(4-cyanopyridin-2-yl)-n-(5-fluoropyridin-3-yl)-5-oxopyrrolidine-2-carboxamide
43. Ac-32624
44. As-35058
45. Hy-18767
46. A14386
47. D11090
48. A900315
49. Q27895417
50. (2s)-1-(4-cyano-2-pyridinyl)-5-oxo-l-prolyl-2-(2-chloroph)-n-(3,3-difluorocyclobutyl)-n2-(5-fluoro-3-pyridinyl)-glycinamide
51. (s)-n-((s)-1-(2-chlorophenyl)-2-((3,3-difluorocyclobutyl)amino)-2-oxoethyl)-1-(4-cyanopyridin-2-yl)-n-(5-fluoropyridin-3-yl)-5-oxopyrrolidine-2-carboxamide;ag-120
52. N-{(1s)-1-(2-chlorophenyl)-2-[(3,3-difluorocyclobutyl)amino]-2-oxoethyl}-1-(4-cyanopyridin-2-yl)-n-(5-fluoropyridin-3-yl)-5-oxo-l-prolinamide
| Molecular Weight | 583.0 g/mol |
|---|---|
| Molecular Formula | C28H22ClF3N6O3 |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 7 |
| Exact Mass | 582.1394008 g/mol |
| Monoisotopic Mass | 582.1394008 g/mol |
| Topological Polar Surface Area | 119 Ų |
| Heavy Atom Count | 41 |
| Formal Charge | 0 |
| Complexity | 1050 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Ivosidenib is approved for use in the treatment of relapsed or refractory AML with a susceptible IDH1 mutation as detected by an FDA-approved test.
FDA Label
Treatment of all conditions included in the category of malignant neoplasms (except central nervous system tumours, haematopoietic and lymphoid tissue neoplasms), Treatment of malignant neoplasms of the central nervous system
Treatment of acute myeloid leukaemia
Many cancers undergo missense mutations of their IDH1 gene leading to substitution of arginine 132 residue of the IDH1 enzyme [A35629. This substitution leads to reduced production of the normal carboxylic acid cycle metabolite -ketoglutarate (-KG) in favor of a new metabolite, D-2-hydroxyglutarate (D-2HG) which reaches levels of 50-100 fold that of wild type cells. D-2HG is a weak competitor to -KG, inhibiting aKG-dependent dioxygenases, and is present . These dioxygenases include several histone demethylases. This leads to hypermethylation of histones, a dominant feature of AML, which is associated with less expression of cell-differentiation genes. Furthermore, methylation sensitive insulators can no longer regulate the activation of oncogenes when histones are hypermethylated. In AML this hypermethylation is known to disrupt hematopoietic differentiation. Ivosidenib reduces the production of D-2HG, relieving the inhibition of histone demethylases and restoring normal methylation conditions. This restores cell differentiation and oncogene regulation leading to regression of the cancer.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01X - Other antineoplastic agents
L01XX - Other antineoplastic agents
L01XX62 - Ivosidenib
Absorption
Ivosidenib has a Tmax of 3 hours following oral administration of a 2 250 mg tablets (total 500 mg). When given with a high-fat meal, Cmax increases by 98% and AUC by 25%. The AUC and Cmax increase in a less than dose-proportional manner in the range of 200-1200 mg daily. Accumulation ratios have been determined to be 1.9 for AUC and 1.5 for Cmax over the course of one month. Steady state is known to be reached within 14 days of daily administration.
Route of Elimination
Following oral administration, 77% of Ivosidenib is eliminated in the feces with 67% present as the parent drug. 17% is excreted in the urine with 10% as the parent drug. No clinically meaningful effects on elimination have been observed with mild to moderate renal impairment or mild hepatic impairment. Changes in patients with severe renal impairment or moderate too severe hepatic impairment have not been investigated.
Volume of Distribution
Ivosidenib has a mean apparent Vd of 234 L at steady-state.
Clearance
Ivosidenib has an apparent clearance rate of 4.3 L/h
Over 92% of Ivosidenib is present in circulation as the parent drug. Metabolism occurs primarily through CYP3A4 with some contribution from N-dealkylation and hydrolysis.
Ivosidenib has a terminal half-life of 93 h.
Ivosidenib is a reversible inhibitor of IDH1 which is non-competitive with respect to the cofactor NADH. It binds to many different 132-substituted IDH1 mutants as well as the wild type enzyme. It is considered to be a slow-binder of the wild type enzyme and binds to mutant enzymes at lower concentrations, both of which may contribute its selectivity. Ivosidenib has not been observed to inhibit any form of IDH2 at micromolar concentrations.