


1. 1353550-13-6
2. Hm61713
3. Bi 1482694
4. 1802181-20-9
5. Olmutinib [inn]
6. Hm-61713
7. Olmutinib [who-dd]
8. Bi-1482694
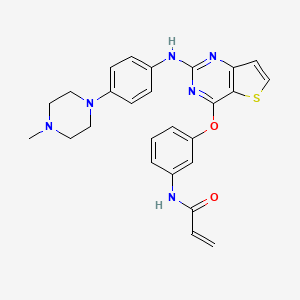
9. N-[3-[2-[4-(4-methylpiperazin-1-yl)anilino]thieno[3,2-d]pyrimidin-4-yl]oxyphenyl]prop-2-enamide
10. Chl9b67l95
11. N-(3-((2-((4-(4-methylpiperazin-1-yl)phenyl)amino)thieno[3,2-d]pyrimidin-4-yl)oxy)phenyl)acrylamide
12. N-{3-[(2-{[4-(4-methylpiperazin-1-yl)phenyl]amino}thieno[3,2-d]pyrimidin-4-yl)oxy]phenyl}prop-2-enamide
13. 2-propenamide, N-(3-((2-((4-(4-methyl-1-piperazinyl)phenyl)amino)thieno(3,2-d)pyrimidin-4-yl)oxy)phenyl)-
14. N-(3-((2-(4-(4-methylpiperazin-1-yl)anilino)thieno(3,2-d)pyrimidin-4-yl)oxy)phenyl)prop-2-enamide
15. 1938072-69-5
16. 2-propenamide, N-[3-[[2-[[4-(4-methyl-1-piperazinyl)phenyl]amino]thieno[3,2-d]pyrimidin-4-yl]oxy]phenyl]-
17. Hm-61713 (bi-1482694)
18. Hm 61713
19. Hm71224;olmutinib
20. Olmutinib (usan/inn)
21. Olmutinib [usan:inn]
22. Olmutinib [usan]
23. Olmutinib(hm-71224)
24. Unii-chl9b67l95
25. Gtpl9196
26. Chembl3786343
27. Schembl14914931
28. Ex-a879
29. Dtxsid001319119
30. Bcp16733
31. Bdbm50160871
32. Mfcd29918158
33. Nsc792848
34. Nsc800980
35. S8294
36. Akos030632791
37. Zinc198970879
38. Ccg-269594
39. Cs-5413
40. Db13164
41. Nsc-792848
42. Nsc-800980
43. Sb19797
44. Example 1 [us20130116213]
45. Ac-30225
46. Ac-35418
47. As-74850
48. Hm 71224
49. Hy-19730
50. Bi1482694
51. Ds-015877
52. Ft-0700176
53. Olmutinib (hm61713, Bi 1482694)
54. J3.435.413h
55. D10859
56. W12116
57. Hm61713, Bi 1482694
58. A900418
59. J-690142
60. Q27088175
61. 4-{[4-({[(1r,2s)-2-phenylcyclopropyl]amino}methyl)-1-piperidinyl]methyl}benzoic Acid
62. Bi1482694;bi 1482694;bi-1482694;hm 61713;hm-61713;hm61713
63. N-(3-(2-(4-(4 Methylpiperazin-1-yl)phenylamino)thieno[3,2-d]pyrimidine-4-yloxy)phenyl)acrylamide
64. N-(3-(2-(4-(4-methylpiperazin-1-yl)phenylamino)thieno[3,2-d]pyrimidine-4-yloxy)phenyl)acrylamide
65. N-(3-{2-[4-(4-methyl-piperazin-1-yl)-phenylamino]-thieno[3,2-d]pyrimidin-4-yloxy}-phenyl)-acrylamide
| Molecular Weight | 486.6 g/mol |
|---|---|
| Molecular Formula | C26H26N6O2S |
| XLogP3 | 4.7 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Exact Mass | 486.18379527 g/mol |
| Monoisotopic Mass | 486.18379527 g/mol |
| Topological Polar Surface Area | 111 Ų |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 712 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For use in treatment of metastatic T790M mutation positive non-small cell lung cancer.
Olmutinib selectively and irreversibly binds and inhibits epidermal growth factor receptors (EGFR) with the T790M activating mutation. EGFRs are frequently over-expressed in lung cancer and contribute to activation of the phosphoinositide 3-kinase and mitogen activated protein kinase pathways which both promote cell survival and proliferation. By inhibiting EGFR activation, olmutinib attenuates the activation of these tumor promoting pathways.
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01E - Protein kinase inhibitors
L01EB - Epidermal growth factor receptor (egfr) tyrosine kinase inhibitors
L01EB06 - Olmutinib
Absorption
tmax of 3-4h with oral administration.
Route of Elimination
Data not yet available.
Volume of Distribution
Data not yet available.
Clearance
Data not yet available.
Data not yet available.
8-11h.
Olmutinib covalently binds a cysteine residue near the kinase domain of mutant EGFRs to prevent phosphorylation of the receptor. This inhibits receptor signalling as phosphorylation is necessary for recruitment of signalling cascade proteins.