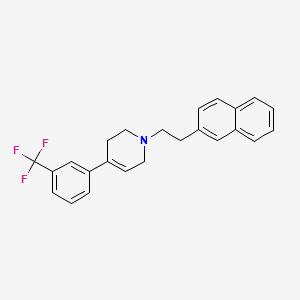

1. 1-(2-(naphth-2-yl)ethyl)-4-(3-trifluoromethylphenyl)-1,2,5,6-tetrahydropyridine Hydrochloride
2. 1-(2-(naphth-2-yl)ethyl)-4-(trifluoromethylphenyl)-1,2,5,6-tetrahydropyridine Hydrochloride
3. Sr 57746a
4. Sr-57746a
5. Sr57746a
6. Xaliproden Hydrochloride
1. 135354-02-8
2. Sr 57746
3. Xaliproden (usan)
4. V8ql94knqo
5. Pyridine, 1,2,3,6-tetrahydro-1-(2-(2-naphthalenyl)ethyl)-4-(3-(trifluoromethyl)phenyl)-
6. Chebi:48520
7. 1,2,3,6-tetrahydro-1-(2-(2-naphthyl)ethyl)-4-(alpha,alpha,alpha-trifluoro-m-tolyl)pyridine
8. Sr-57746
9. Xaliproden [usan]
10. 1,2,3,6-tetrahydro-1-(2-(2-naphthalenyl)ethyl)-4-(3-(trifluoromethyl)phenyl)-pyridine
11. 1-(2-(2-naphthyl)ethyl)-4-(3-trifluoromethylphenyl)-1,2,3,6-tetrahydropyridine
12. 1-[2-(2-naphthyl)ethyl]-4-[3-(trifluoromethyl)phenyl]-1,2,3,6-tetrahydropyridine
13. Ncgc00167574-01
14. Unii-v8ql94knqo
15. Xaliproden [usan:inn:ban]
16. 1-[2-(2-naphthyl)ethyl]-4-(3-trifluoromethylphenyl)-1,2,3,6-tetrahydropyridine
17. Xaliproden [mi]
18. Xaliproden [inn]
19. 1-(2-naphthalen-2-ylethyl)-4-[3-(trifluoromethyl)phenyl]-3,6-dihydro-2h-pyridine
20. Xaliproden [who-dd]
21. Schembl137094
22. Chembl1512580
23. Dtxsid9048306
24. Bdbm82363
25. Zinc577115
26. Pdsp1_000054
27. Pdsp2_000054
28. Db06393
29. Sr 57746 A
30. Ncgc00167574-02
31. Cas_90494-79-4
32. Db-063077
33. Ft-0736660
34. D06327
35. L001401
36. Brd-k88358234-003-01-4
37. Q12745720
38. 1-(2-naphthalen-2-ylethyl)-4-[3-(trifluoromethyl)phenyl]-1,2,3,6-tetrahydropyridine
39. 1-[2(naphth-2-yl)ethyl]-4-(3-trifluoromethylphenyl)-1,2,5,6-tetrahydropyridine
40. 1-[2-(naphth-2-yl)ethyl]-4-(3-trifluoromethylphenyl)-1,2,3,6-tetrahydropyridine
| Molecular Weight | 381.4 g/mol |
|---|---|
| Molecular Formula | C24H22F3N |
| XLogP3 | 6.2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 381.17043419 g/mol |
| Monoisotopic Mass | 381.17043419 g/mol |
| Topological Polar Surface Area | 3.2 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 541 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Investigated for use/treatment in amyotrophic lateral sclerosis (ALS) and alzheimer's disease.
Serotonin 5-HT1 Receptor Agonists
Endogenous compounds and drugs that specifically stimulate SEROTONIN 5-HT1 RECEPTORS. Included under this heading are agonists for one or more of the specific 5-HT1 receptor subtypes. (See all compounds classified as Serotonin 5-HT1 Receptor Agonists.)
N - Nervous system
N07 - Other nervous system drugs
N07X - Other nervous system drugs
N07XX - Other nervous system drugs
N07XX03 - Xaliproden
Xaliproden is an orally-active, synthetic, non-peptidic 5-hydroxytryptamine (5-HT) 1A receptor agonist with neurotrophic and neuroprotective activities. Although its mechanism of action is not fully understood, xaliproden appears to either mimic the effects of neurotrophins or stimulate their synthesis, thereby stimulating neuronal cell differentiation and proliferation and inhibiting neuronal cell death. The neuroprotective effect of this agent involves the activation of MAP kinase pathways via stimulation of the 5-HT1A receptor.