
Synopsis
Synopsis
0
VMF
0
South Africa

0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
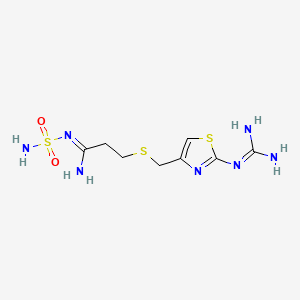

1. Famotidine
2. Famotidine Hydrochloride
3. Mk 208
4. Mk-208
5. Mk208
6. Ym 11170
7. Ym-11170
8. Ym11170
1. Famotidine
2. 76824-35-6
3. Gastridin
4. Famodil
5. Pepdine
6. Pepdul
7. Pepcidine
8. Famoxal
9. Ganor
10. Pepcidac
11. Amfamox
12. Gastropen
13. Famosan
14. Fluxid
15. Gaster
16. Lecedil
17. Motiax
18. Muclox
19. Pepcid Ac
20. Quamatel
21. Ym-11170
22. Pepcid Rpd
23. Fadul
24. Mk-208
25. Famotidine (pepcid)
26. 3-[({2-[(diaminomethylidene)amino]-1,3-thiazol-4-yl}methyl)sulfanyl]-n-sulfamoylpropanimidamide
27. L 643341
28. Mk 208
29. Chebi:4975
30. Ym 11170
31. Nsc-757810
32. 3-(((2-((diaminomethylene)amino)thiazol-4-yl)methyl)thio)-n'-sulfamoylpropanimidamide
33. Xufqphaneapemj-uhfffaoysa-
34. 3-(((2-guanidinothiazol-4-yl)methyl)thio)-n-sulfamoylpropanimidamide
35. (1-amino-3-(((2-((diaminomethylene)amino)-4-thiazolyl)methyl)thio)propylidene)sulfamide
36. Pepcid Ac Gelcaps
37. Smr000058961
38. Pepcid (tn)
39. Chembl902
40. 3-[[[2-[(aminoiminomethyl)amino]-4-thiazolyl]methyl]thio]-n-(aminosulfonyl)propanimidamide
41. Sr-05000001440
42. 5qzo15j2z8
43. Ncgc00015446-05
44. Fo9
45. Propanimidamide, N'-(aminosulfonyl)-3-(((2-((diaminomethylene)amino)-4-thiazolyl)methyl)thio)-
46. Prestwick_212
47. Cas-76824-35-6
48. Fluxid (tn)
49. Mfcd00079297
50. Hs-0054
51. Ym-1170
52. Famotidine [mi]
53. 3-(2-guanidinothiazol-4-ylmethylthio)-n1-sulfamoylpropionamide
54. Famotidine [inn]
55. Famotidine [jan]
56. Prestwick2_000104
57. Prestwick3_000104
58. Famotidine [hsdb]
59. Famotidine [usan]
60. Lopac-f-6889
61. F0530
62. Schembl972
63. Schembl974
64. Famotidine [vandf]
65. F 6889
66. Famotidine [mart.]
67. Famotidine [usp-rs]
68. Famotidine [who-dd]
69. N'-(aminosulfonyl)-3-([2-(diaminomethyleneamino)-4-thiazolyl]methylthio)propanamidine
70. Lopac0_000497
71. Bspbio_000088
72. Mls000028583
73. Mls000758205
74. Mls001423994
75. Bidd:gt0759
76. Bpbio1_000098
77. Famotidine (jp17/usp/inn)
78. Famotidine [orange Book]
79. Bdbm22891
80. Cid_5702160
81. Famotidine [ep Monograph]
82. Famotidine [usp Impurity]
83. Famotidine [usp Monograph]
84. Hms1568e10
85. Hms2051a07
86. Hms2089i12
87. Hms2095e10
88. Hms2231i22
89. Hms3261d15
90. Hms3712e10
91. Hms3884j03
92. Hy-b0377
93. Tox21_500497
94. Bdbm50103514
95. Pdsp1_000398
96. Pdsp2_000396
97. S2078
98. Stk527689
99. Akos005460541
100. Akos015994617
101. Ccg-100767
102. Ccg-220104
103. Ccg-221801
104. Nc00017
105. Sdccgsbi-0050481.p004
106. 3-[[2-(diaminomethylideneamino)-1,3-thiazol-4-yl]methylsulfanyl]-n'-sulfamoylpro
107. Propanimidamide, 3-[[[2-[aminoiminomethyl)amino]-4-thiazoyl]methyl]thio]-n-(aminosulfonyl)
108. Ncgc00015446-01
109. Ncgc00015446-02
110. Ncgc00015446-03
111. Ncgc00015446-04
112. Ncgc00015446-06
113. Ncgc00015446-08
114. Ncgc00018276-01
115. Ncgc00018276-02
116. Ncgc00093899-01
117. Ncgc00093899-02
118. Ncgc00093899-03
119. Ncgc00188952-01
120. Ncgc00261182-01
121. (1z)-3-[({2-[(diaminomethylidene)amino]-1,3-thiazol-4-yl}methyl)sulfanyl]-n'-sulfamoylpropanimidamide
122. (1z)-n'-(aminosulfonyl)-3-[({2-[(diaminomethylidene)amino]-1,3-thiazol-4-yl}methyl)thio]propanimidamide
123. Ac-11713
124. Ac-31723
125. Pepcid Complete Component Famotidine
126. Sbi-0050481.p003
127. Eu-0100497
128. Famotidine Component Of Pepcid Complete
129. D00318
130. Ab00383032_11
131. 824f356
132. A838850
133. A899959
134. L003830
135. L013386
136. Sr-01000075883
137. Sr-01000075883-1
138. Sr-05000001440-1
139. Sr-05000001440-2
140. Famotidine, British Pharmacopoeia (bp) Reference Standard
141. Famotidine, European Pharmacopoeia (ep) Reference Standard
142. Famotidine, United States Pharmacopeia (usp) Reference Standard
143. 3-[(2-guanidinothiazol-4-yl)methylsulfanyl]-n-sulfamoyl-propanamidine
144. Famotidine, Pharmaceutical Secondary Standard; Certified Reference Material
145. 3-((((2-(diaminomethylene)amino)-4-thiazolyl)methyl)thio)-n-sulfamoylpropionamidine
146. 3-(2-(diaminomethyleneamino)-1,3-thiazol-4-yl)methylthio)-n'- Sulfamoylpropionamidine
147. 3-[2-(diaminomethyleneamino] Thiazol-4-ylmethylthio]-n-sulphamoyl Propionamidine
148. Famotidine For System Suitability, European Pharmacopoeia (ep) Reference Standard
149. 3-[({2-[(diaminomethylene)amino]-1,3-thiazol-4-yl}methyl)sulfanyl]-n'-sulfamoylpropanimidamide
150. 3-[({2-[(diaminomethylidene)amino]-1,3-thiazol-4-yl}methyl)sulfanyl]-n''-sulfamoylpropanimidamide
| Molecular Weight | 337.5 g/mol |
|---|---|
| Molecular Formula | C8H15N7O2S3 |
| XLogP3 | -0.6 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Exact Mass | 337.04493627 g/mol |
| Monoisotopic Mass | 337.04493627 g/mol |
| Topological Polar Surface Area | 238 Ų |
| Heavy Atom Count | 20 |
| Formal Charge | 0 |
| Complexity | 469 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 10 | |
|---|---|
| Drug Name | Famotidine |
| Active Ingredient | Famotidine |
| Dosage Form | Injectable; Tablet; Tablet, chewable; For suspension; Suspension |
| Route | injection; Injection; Oral |
| Strength | 40mg/5ml; 10mg; 10mg/ml; 40mg; 20mg |
| Market Status | Tentative Approval; Over the Counter; Prescription |
| Company | Navinta; Ranbaxy; Wockhardt; Bedford; Ivax Sub Teva Pharms; Marsam Pharms; Fresenius Kabi Usa; Hi-tech Pharma; Hikma Maple; Teva; Apotex; Perrigo; Alembic Pharms; Lupin; Dr Reddys Labs; Carlsbad; Agila Speclts; Mylan; Novel Labs |
| 2 of 10 | |
|---|---|
| Drug Name | Famotidine preservative free |
| PubMed Health | Famotidine |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in PEPCID (famotidine) is a histamine H2-receptor antagonist. Famotidine is N'-(aminosulfonyl)-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]propanimidamide. The empirical formula of famotidine is C8H15N7O2S3 and it.. |
| Active Ingredient | Famotidine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 10mg/ml |
| Market Status | Prescription |
| Company | Bedford; Fresenius Kabi Usa; Hikma Maple; Bedford Labs; Agila Speclts |
| 3 of 10 | |
|---|---|
| Drug Name | Famotidine preservative free in plastic container |
| PubMed Health | Famotidine (By mouth) |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Active Ingredient | Famotidine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 0.4mg/ml |
| Market Status | Prescription |
| Company | Baxter Hlthcare |
| 4 of 10 | |
|---|---|
| Drug Name | Pepcid |
| PubMed Health | Famotidine |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in Famotidine Injection, USP is a histamine H2-receptor antagonist. Famotidine is [1-Amino-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]propylidene] sulfamide. Its structural formula is:C8H15N7O2S3 MW 337.4... |
| Active Ingredient | Famotidine |
| Dosage Form | Tablet; For suspension |
| Route | Oral |
| Strength | 40mg/5ml; 40mg; 20mg |
| Market Status | Prescription |
| Company | Marathon Pharms; Salix Pharms |
| 5 of 10 | |
|---|---|
| Drug Name | Pepcid ac |
| Active Ingredient | Famotidine |
| Dosage Form | Tablet; Tablet, chewable |
| Route | Oral |
| Strength | 10mg; 20mg |
| Market Status | Over the Counter |
| Company | Mcneil Cons |
| 6 of 10 | |
|---|---|
| Drug Name | Famotidine |
| Active Ingredient | Famotidine |
| Dosage Form | Injectable; Tablet; Tablet, chewable; For suspension; Suspension |
| Route | injection; Injection; Oral |
| Strength | 40mg/5ml; 10mg; 10mg/ml; 40mg; 20mg |
| Market Status | Tentative Approval; Over the Counter; Prescription |
| Company | Navinta; Ranbaxy; Wockhardt; Bedford; Ivax Sub Teva Pharms; Marsam Pharms; Fresenius Kabi Usa; Hi-tech Pharma; Hikma Maple; Teva; Apotex; Perrigo; Alembic Pharms; Lupin; Dr Reddys Labs; Carlsbad; Agila Speclts; Mylan; Novel Labs |
| 7 of 10 | |
|---|---|
| Drug Name | Famotidine preservative free |
| PubMed Health | Famotidine |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in PEPCID (famotidine) is a histamine H2-receptor antagonist. Famotidine is N'-(aminosulfonyl)-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]propanimidamide. The empirical formula of famotidine is C8H15N7O2S3 and it.. |
| Active Ingredient | Famotidine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 10mg/ml |
| Market Status | Prescription |
| Company | Bedford; Fresenius Kabi Usa; Hikma Maple; Bedford Labs; Agila Speclts |
| 8 of 10 | |
|---|---|
| Drug Name | Famotidine preservative free in plastic container |
| PubMed Health | Famotidine (By mouth) |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Active Ingredient | Famotidine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 0.4mg/ml |
| Market Status | Prescription |
| Company | Baxter Hlthcare |
| 9 of 10 | |
|---|---|
| Drug Name | Pepcid |
| PubMed Health | Famotidine |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in Famotidine Injection, USP is a histamine H2-receptor antagonist. Famotidine is [1-Amino-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]propylidene] sulfamide. Its structural formula is:C8H15N7O2S3 MW 337.4... |
| Active Ingredient | Famotidine |
| Dosage Form | Tablet; For suspension |
| Route | Oral |
| Strength | 40mg/5ml; 40mg; 20mg |
| Market Status | Prescription |
| Company | Marathon Pharms; Salix Pharms |
| 10 of 10 | |
|---|---|
| Drug Name | Pepcid ac |
| Active Ingredient | Famotidine |
| Dosage Form | Tablet; Tablet, chewable |
| Route | Oral |
| Strength | 10mg; 20mg |
| Market Status | Over the Counter |
| Company | Mcneil Cons |
Anti-Ulcer Agents; Histamine H2 Antagonists
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Famotidine is currently the drug of choice for initial treatment and maintenance therapy in most patients with uncomplicated gastric or duodenal ulcer. ... A single bedtime dose of famotidine 40 mg is as efficatious as previously recommended multidose regimens and increases compliance.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 892
Histamine H2-receptor antagonists are indicated in the short-term treatment of active duodenal ulcer. They are also indicated (at reduce dosage) in the prevention of duodenal ulcer recurrence in selected patients. /Histamine H2-receptor antagonists; Included in US product labeling/
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 1611
Famotidine ... /is/ indicated in the short-term treatment of active benign gastric ulcer. /Included in US product labeling/
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 1611
For more Therapeutic Uses (Complete) data for FAMOTIDINE (12 total), please visit the HSDB record page.
Although appropriate studies on the relationship of age to the effects of these medicines /cimetidine, famotidine, and ranitidine/ have not been performed in the geriatric population, no geriatrics-specific problems have been documented to date. However, confusion is more likely to occur in elderly patients with impaired hepatic or renal function.
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 1613
Adverse nervous system effects (eg, headache, dizziness) and GI effects (eg, constipation, diarrhea) occur most frequently during famotidine therapy. Although adverse effects of the drug generally are not severe, discontinuance of famotidine therapy has been necessary in up to 14% of patients. Adverse effects generally are similar when famotidine is administered orally or IV.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2151
Fever, hypertension, flushing, musculoskeletal pain, arthralgia, and tinnitus have been reported in 1% or less of patients receiving famotidine, but a causal relationship to the drug has not been established in many cases. An acute episode of gout occurred in one patient during therapy with the drug.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2151
Leukocytosis, leukopenia, neutropenia, pancytopenia, agranulocytosis, eosinophilia, prolonged erythrocyte sedimentation rate (ESR), and thrombocytopenia have occurred rarely in patients receiving famotidine. Changes in serum protein or cholesterol concentrations also have occurred.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2151
For more Drug Warnings (Complete) data for FAMOTIDINE (10 total), please visit the HSDB record page.
Anti-Ulcer Agents
Various agents with different action mechanisms used to treat or ameliorate PEPTIC ULCER or irritation of the gastrointestinal tract. This has included ANTIBIOTICS to treat HELICOBACTER INFECTIONS; HISTAMINE H2 ANTAGONISTS to reduce GASTRIC ACID secretion; and ANTACIDS for symptomatic relief. (See all compounds classified as Anti-Ulcer Agents.)
Histamine H2 Antagonists
Drugs that selectively bind to but do not activate histamine H2 receptors, thereby blocking the actions of histamine. Their clinically most important action is the inhibition of acid secretion in the treatment of gastrointestinal ulcers. Smooth muscle may also be affected. Some drugs in this class have strong effects in the central nervous system, but these actions are not well understood. (See all compounds classified as Histamine H2 Antagonists.)
A - Alimentary tract and metabolism
A02 - Drugs for acid related disorders
A02B - Drugs for peptic ulcer and gastro-oesophageal reflux disease (gord)
A02BA - H2-receptor antagonists
A02BA03 - Famotidine
All H2-receptor antagonists are distributed in breast milk and cerebral spinal fluid. /Histamine H2-receptor antagonists/
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 1612
Distribution of famotidine into human body tissues and fluids has not been fully characterized. The apparent volume of distribution of the drug is reported to be 1.1-1.4 l/kg in adults and does not appear to be altered substantially in patients with renal dysfunction. Following oral or IV administration in rats, famotidine is widely distributed, appearing in highest concentrations in the kidney, liver, pancreas, and submandibular gland. The drug is 15-20% protein bound.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2149
In rats famotidine appears to distribute only minimally into the CNS, and does not cross the placenta. It is not known whether the drug crosses the placenta in humans. Famotidine is distributed into milk in rats; however, it is not known whether the drug is distributed into milk in humans.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2149
Famotidine is excreted principally in urine via glomerular filtration and tubular secretion. Approximately 25-30 or 65-80% of a dose is excreted unchanged in urine within 24 hours following oral or IV administration, respectively, and approximately 13-49 or 52-82% of a single 40 mg oral or IV dose respectively, is excreted within 72 hours. ... The remainder of an orally administered dose is eliminated in feces.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2149
For more Absorption, Distribution and Excretion (Complete) data for FAMOTIDINE (7 total), please visit the HSDB record page.
Famotidine is metabolized in the liver to famotidine S-oxide (S-famotidine). The metabolite does not appear to inhibit gastric acid secretion. Orally administered famotidine undergoes minimal metabolism on first pass through the liver.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2149
H2-receptor antagonists inhibit basal and nocturnal gastric acid secretion by competitive inhibition of the action of histamine at the histamine H2-receptors of the parietal cells. They also inhibit gastric acid secretion stimulated by food, betazole, pentagastrin, caffeine, insulin, and physiological vagal reflex. /Histamine H2-receptor antagonists/
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 1612
Weak inhibitor of hepatic cytochrome p450 mixed function oxidase system.
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 1612
Famotidine is a competitive H2 receptor antagonist that inhibits basal, overnight, and pentagastrin-stimulated gastric acid secretion. Pharmacologically, it is three times more potent than ranitidine and 20 times more potent than cimetidine.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 902

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?