NDC Code(s) : 0069-0121-01, 0069-0121-02, 0069-0125-01, 0069-0125-02, 0069-0126-01, 0069-0126-02
Packager : Pfizer Laboratories Div Pfizer Inc
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| FamotidineFAMOTIDINE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FamotidineFAMOTIDINE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| FamotidineFAMOTIDINE INJECTION, SOLUTION | ||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
PRINCIPAL DISPLAY PANEL
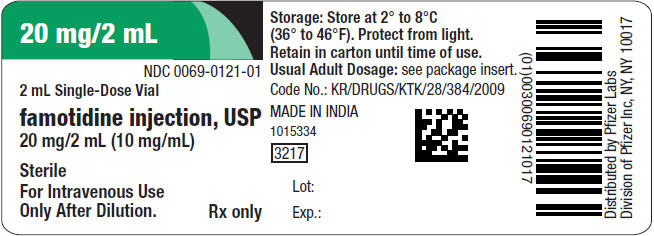
20 mg/2 mL
NDC 0069-0121-01
2 mL Single-Dose Vial
famotidine injection, USP
20 mg/2 mL (10 mg/mL)
Sterile
For Intravenous Use
Only After Dilution.
Rx only
PRINCIPAL DISPLAY PANEL
NDC 0069-0121-02
Contains 25 of NDC 0069-0121-01
Rx only
25 x 2 mL Single-Dose Vials
famotidine injection, USP
20 mg/2 mL (10 mg/mL)
Sterile
For Intravenous Use Only After Dilution.
Pfizer Injectables
20 mg/2 mL

PRINCIPAL DISPLAY PANEL
NDC 0069-0125-01
4 mL Multi-dose Vial
Famotidine
Injection, USP
40 mg/4 mL
(10 mg/mL)
For Intravenous Use
Only After Dilution.
Rx only
Sterile

PRINCIPAL DISPLAY PANEL
NDC 0069-0125-02
Contains 10 of NDC 0069-0125-01
Rx only
10 x 4 mL Multi-dose Vial
Famotidine Injection, USP
40 mg/4 mL
(10 mg/mL)
Sterile
For Intravenous Use Only After Dilution.
Pfizer Injectables

PRINCIPAL DISPLAY PANEL
NDC 0069-0126-01
20 mL Multi-dose Vial
Famotidine
Injection, USP
200 mg/20 mL
(10 mg/mL)
Sterile
For Intravenous Use
Only After Dilution.
Rx only

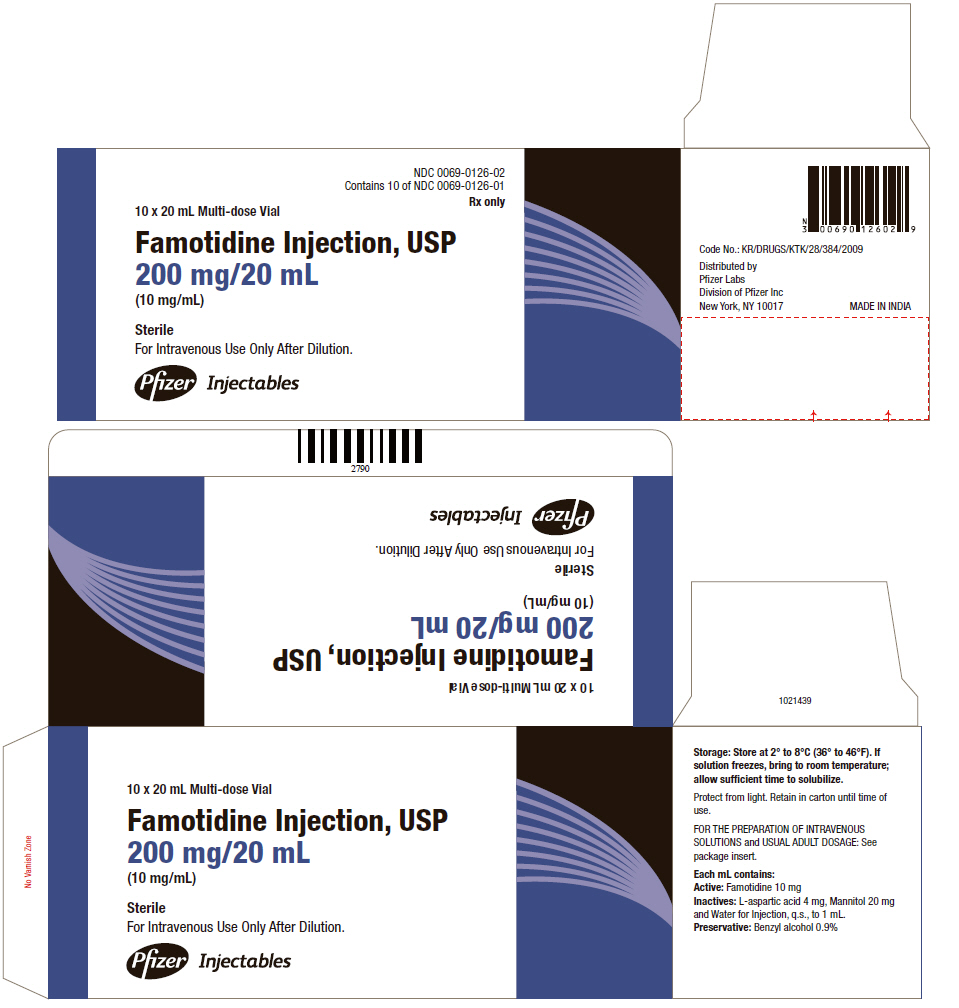
PRINCIPAL DISPLAY PANEL
NDC 0069-0126-02
Contains 10 of NDC 0069-0126-01
Rx only
10 x 20 mL Multi-dose Vial
Famotidine Injection, USP
200 mg/20 mL
(10 mg/mL)
Sterile
For Intravenous Use Only After Dilution.
Pfizer Injectables