NDC Code(s) : 10812-192-01
Packager : Johnson & Johnson Consumer Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Neutrogena Fresh Cooling Sunscreen Avobenzone, Homosalate, Octisalate, Octocrylene, and Oxybenzone AEROSOL, SPRAY | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
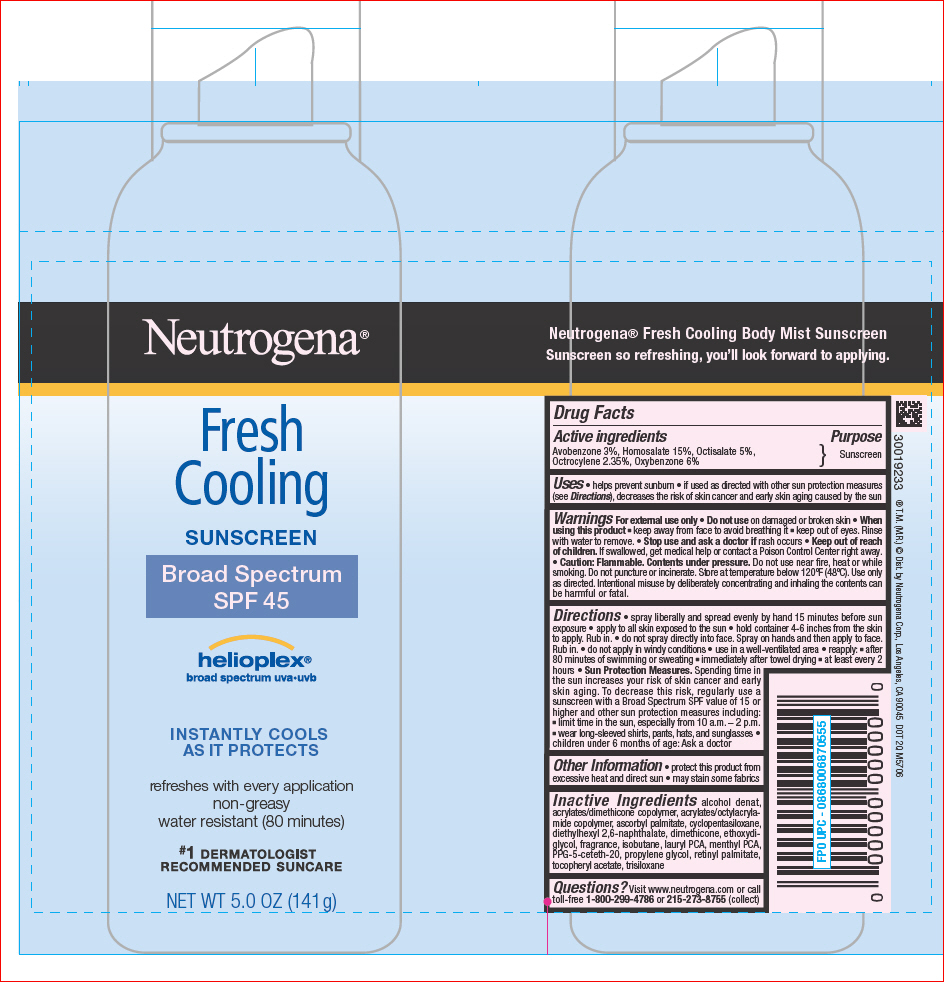
PRINCIPAL DISPLAY PANEL
Neutrogena®
Fresh
Cooling
SUNSCREEN
Broad Spectrum
SPF 45
helioplex®
broad spectrum uva•uvb
INSTANTLY COOLS
AS IT PROTECTS
refreshes with every application
non-greasy
water resistant (80 minutes)
#1 DERMATOLOGIST
RECOMMENDED SUNCARE
NET WT 5.0 OZ (141 g)