NDC Code(s) : 32909-717-01
Packager : E-Z-EM Canada Inc
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| E-Z-Cat DryBarium Sulfate POWDER, FOR SUSPENSION | ||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
PRINCIPAL DISPLAY PANEL
E-Z-CAT
DRY Box
NDC: 32909-717-01
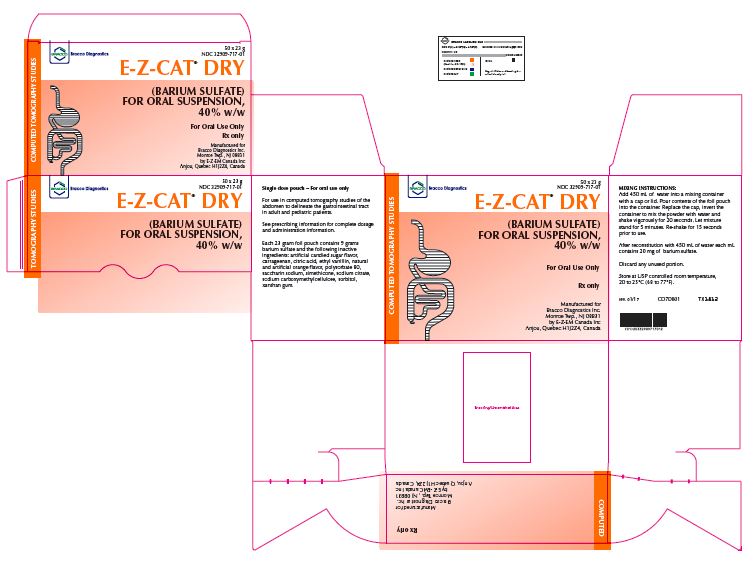
E-Z-CAT DRY Box Labels











