NDC Code(s) : 42507-494-21
Packager : HyVee Inc
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| cough dmdextromethorphan polistirex SUSPENSION, EXTENDED RELEASE | ||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
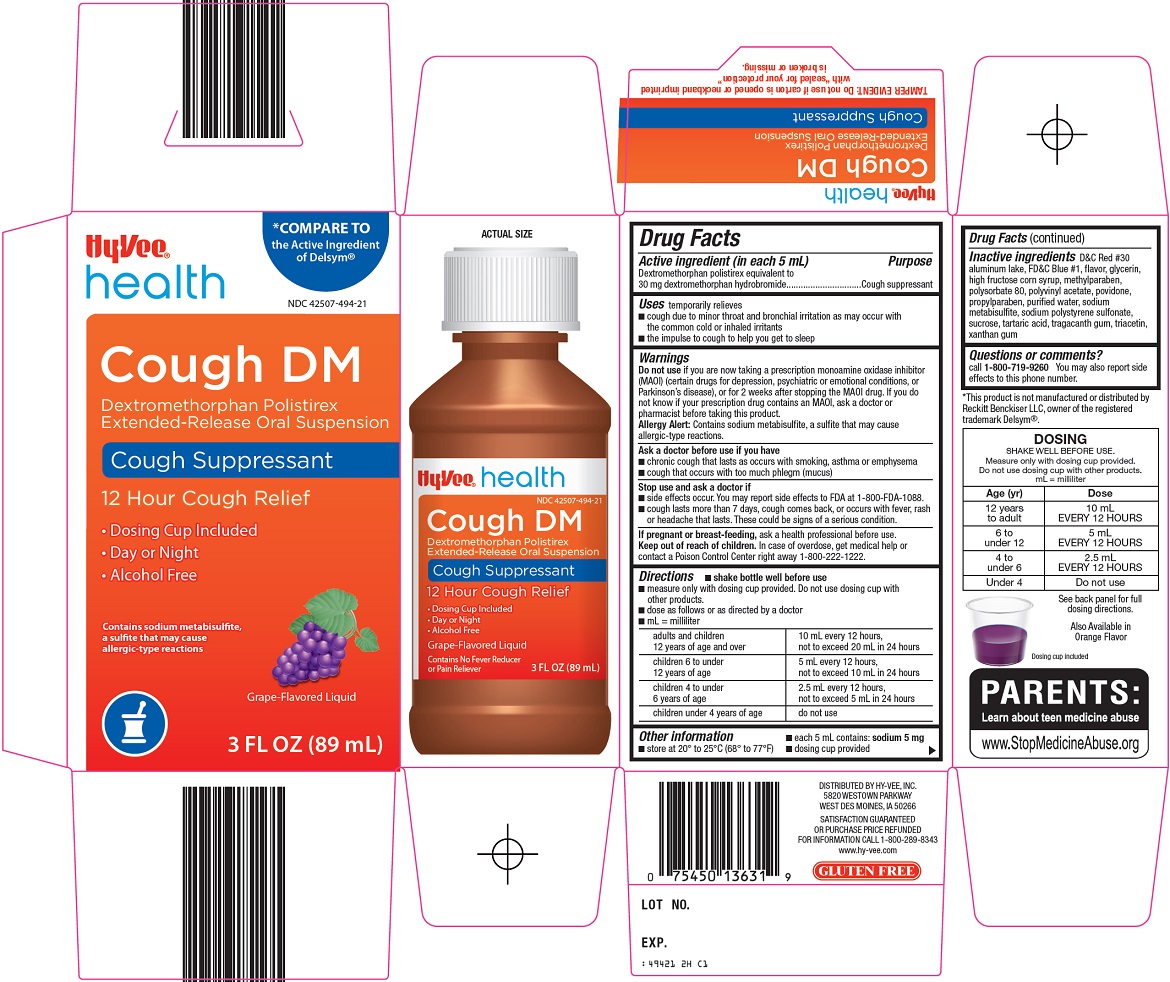
PRINCIPAL DISPLAY PANEL
COMPARE TO the Active Ingredient of Delsym®
Cough DM
Dextromethorphan Polistirex Extended-Release Oral Suspension
Cough Suppressant
12 Hour Cough Relief
Dosing Cup Included
Day or Night
Alcohol Free
Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions
Grape-Flavored Liquid
3 FL OZ (89 mL)