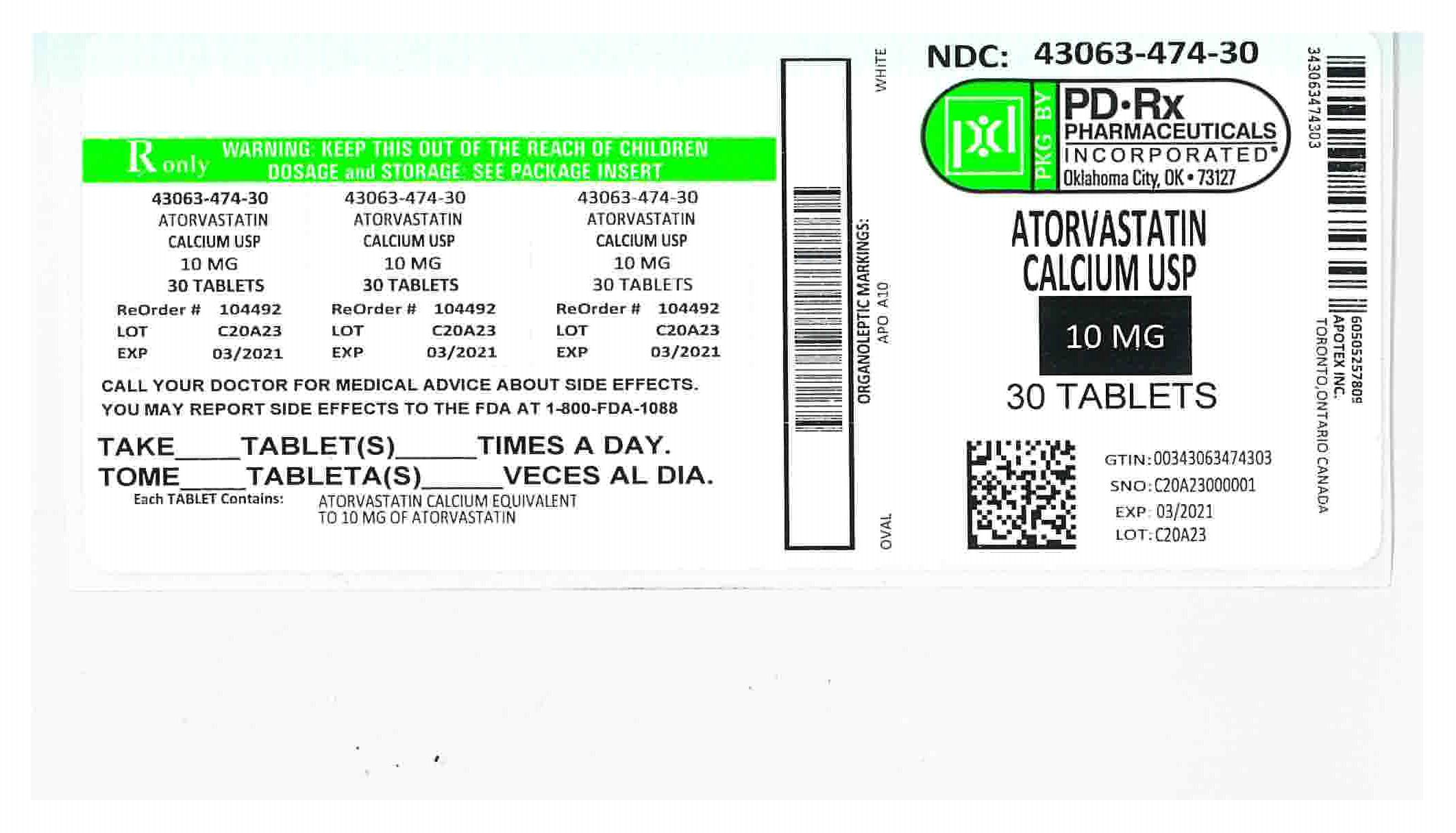
NDC Code(s) : 43063-474-30, 43063-474-90
Packager : PD-Rx Pharmaceuticals, Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| ATORVASTATIN CALCIUMATORVASTATIN CALCIUM TABLET, FILM COATED | ||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| LABELER - PD-Rx Pharmaceuticals, Inc.(156893695) |
| REGISTRANT - PD-Rx Pharmaceuticals, Inc.(156893695) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| PD-Rx Pharmaceuticals, Inc. | 156893695 | repack(43063-474) | |
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 10 mg BOTTLE LABEL
Representative sample of labeling (see HOW SUPPLIED section for complete listing):
Atorvastatin Calcium Tablets
10 mg
Rx