NDC Code(s) : 49967-500-01, 49967-500-02, 49967-500-03, 49967-500-04, 49967-500-05, 49967-500-06, 49967-500-07, 49967-500-08
Packager : L'Oreal USA Products Inc.
Category : HUMAN OTC DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| CeraVe Developed with Dermatologists HealingPetrolatum OINTMENT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LABELER - L'Oreal USA Products Inc.(002136794) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Accupac, Inc. | 071609663 | MANUFACTURE(49967-500) | |
PRINCIPAL DISPLAY PANEL
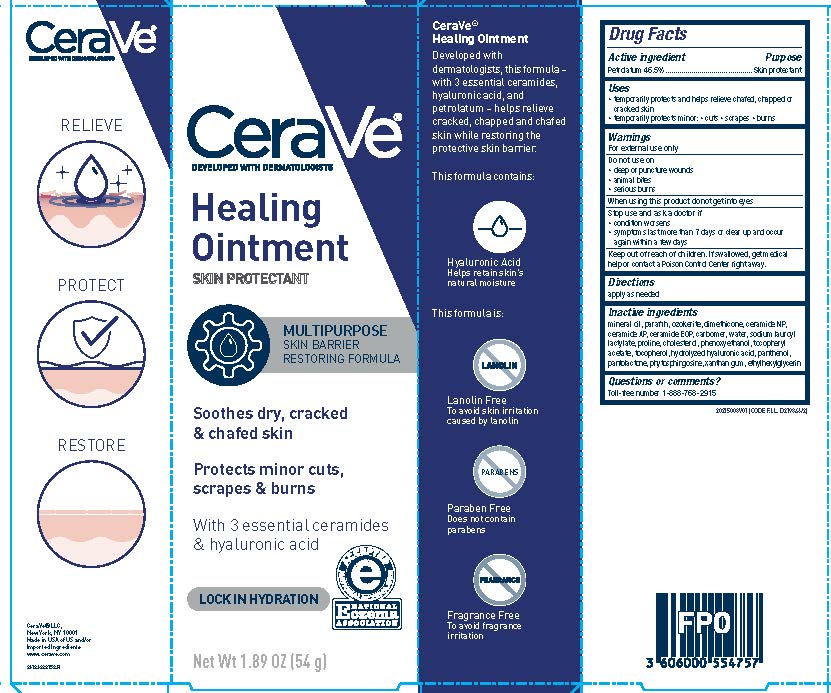
PRINCIPAL DISPLAY PANEL


PRINCIPAL DISPLAY PANEL


PRINCIPAL DISPLAY PANEL











