NDC Code(s) : 65862-596-01, 65862-596-02
Packager : Aurobindo Pharma Limited
Category : Human Prescription Drug Label
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| Clindamycin Palmitate Hydrochloride (Pediatric) Clindamycin Palmitate Hydrochloride (Pediatric) SOLUTION | ||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| LABELER - Aurobindo Pharma Limited(650082092) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Aurobindo Pharma Limited | 918917642 | ANALYSIS(65862-596), MANUFACTURE(65862-596) | |
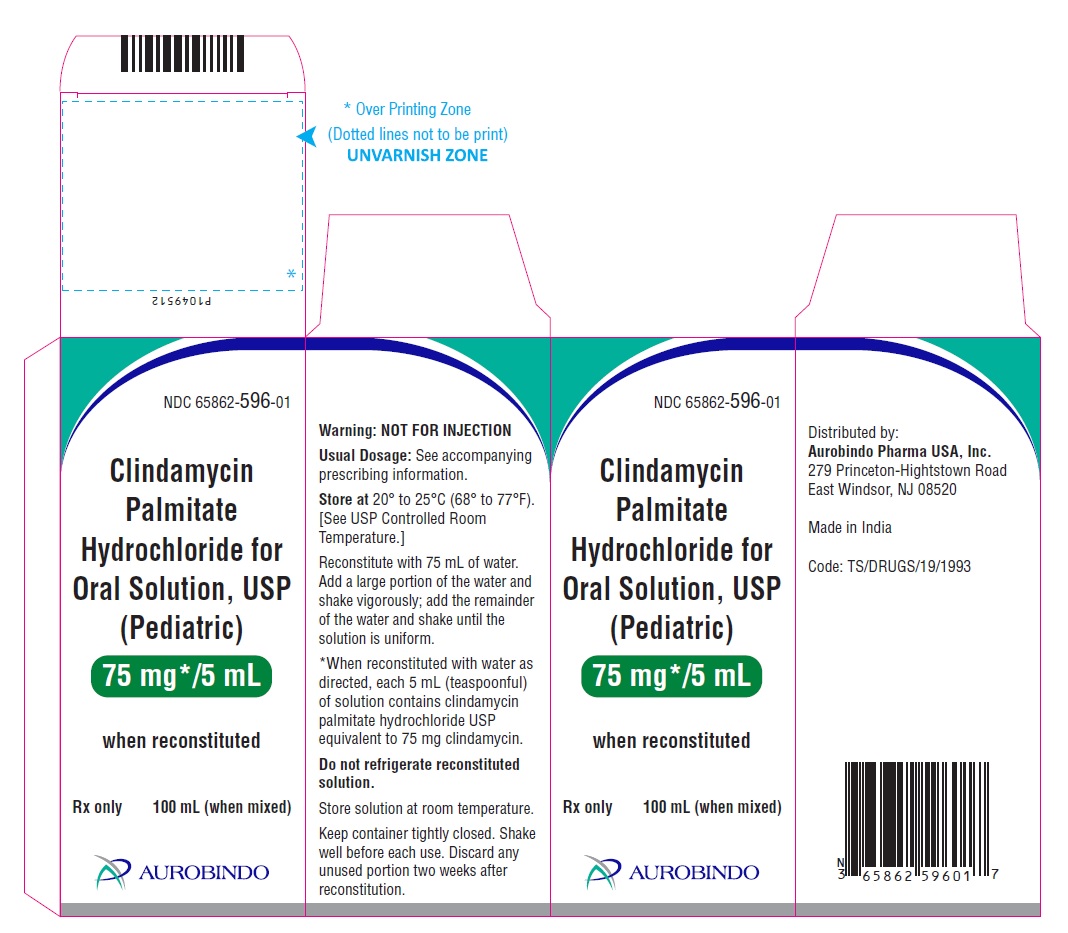
PRINCIPAL DISPLAY PANEL
NDC 65862-596-01
Rx only
Clindamycin Palmitate
Hydrochloride
for Oral Solution, USP (Pediatric)
75 mg*/5 mL
when reconstituted
AUROBINDO 100 mL (when mixed)
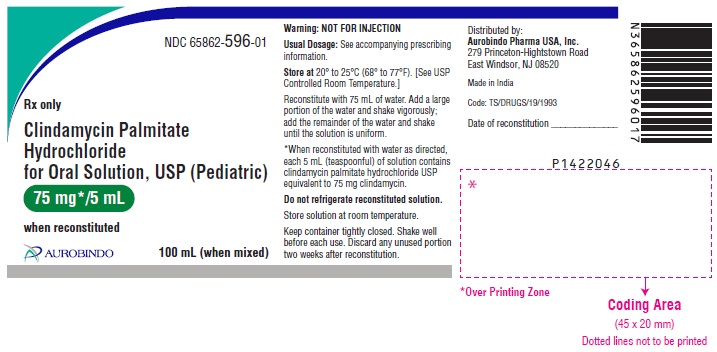
PRINCIPAL DISPLAY PANEL
NDC 65862-596-01
Clindamycin
Palmitate
Hydrochloride for
Oral Solution, USP
(
Pediatric)
75 mg*/5 mL
when reconstituted
Rx only 100 mL (when mixed)
AUROBINDO