NDC Code(s) : 68382-324-01, 68382-324-04, 68382-325-01, 68382-325-04, 68382-326-01, 68382-326-04, 68382-327-01, 68382-327-04, 68382-328-01, 68382-328-04, 68382-329-01, 68382-329-04
Packager : Zydus Pharmaceuticals USA Inc.
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| estradiolestradiol PATCH | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| estradiolestradiol PATCH | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| estradiolestradiol PATCH | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| estradiolestradiol PATCH | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| estradiolestradiol PATCH | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| estradiolestradiol PATCH | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| LABELER - Zydus Pharmaceuticals USA Inc.(156861945) |
| REGISTRANT - ZYDUS NOVELTECH INC, USA(801012530) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Zydus Lifesciences Limited | 918596198 | ANALYSIS(68382-324, 68382-325, 68382-326, 68382-327, 68382-328, 68382-329), MANUFACTURE(68382-324, 68382-325, 68382-326, 68382-327, 68382-328, 68382-329) | |
PRINCIPAL DISPLAY PANEL
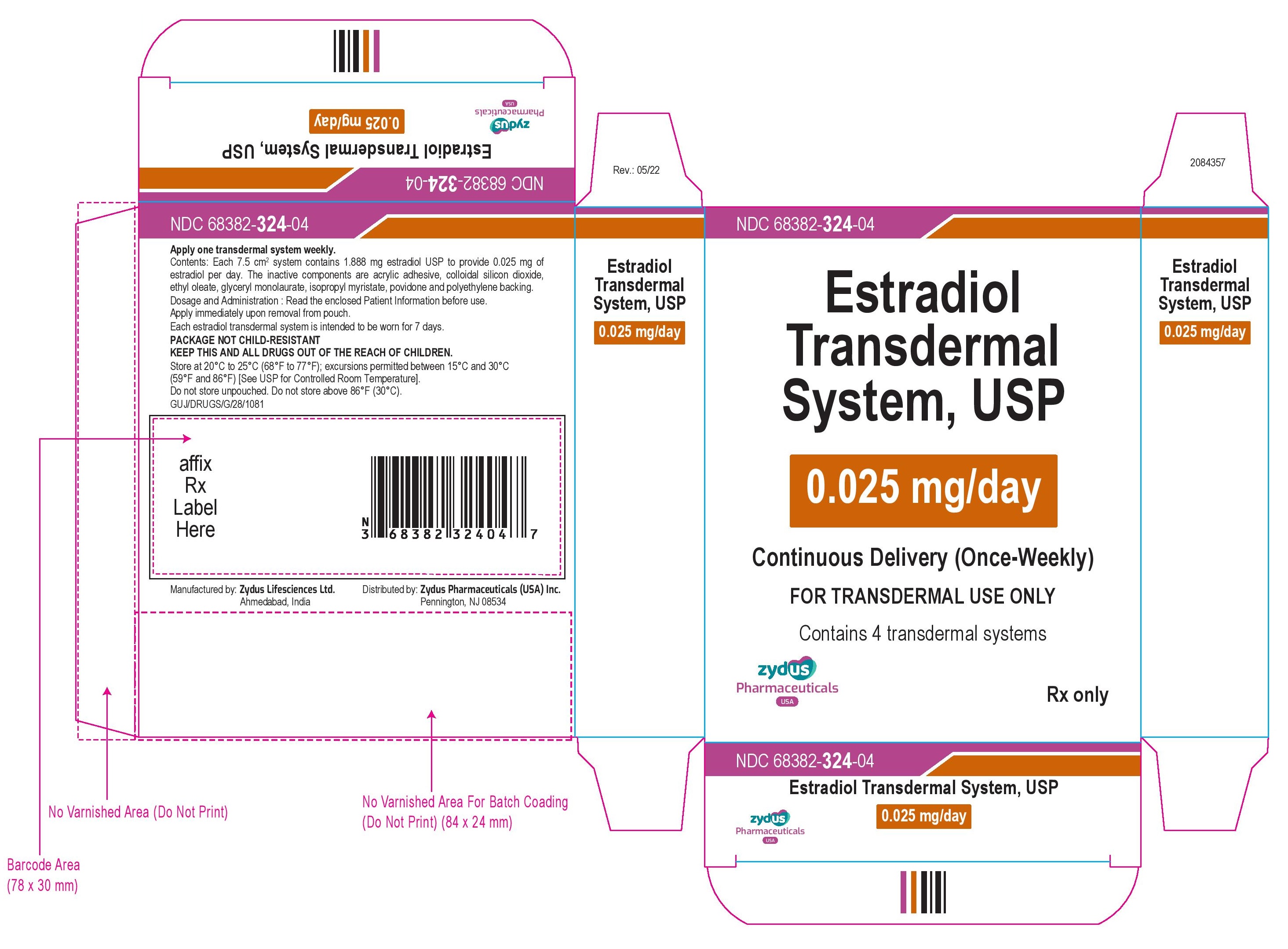
NDC 68382-324-04
Estradiol Transdermal System, USP
0.025 mg/day
Continuous Delivery (Once-Weekly)
FOR TRANSDERMAL USE ONLY
Contains 4 transdermal systems
Rx only
zydus Pharmaceuticals USA
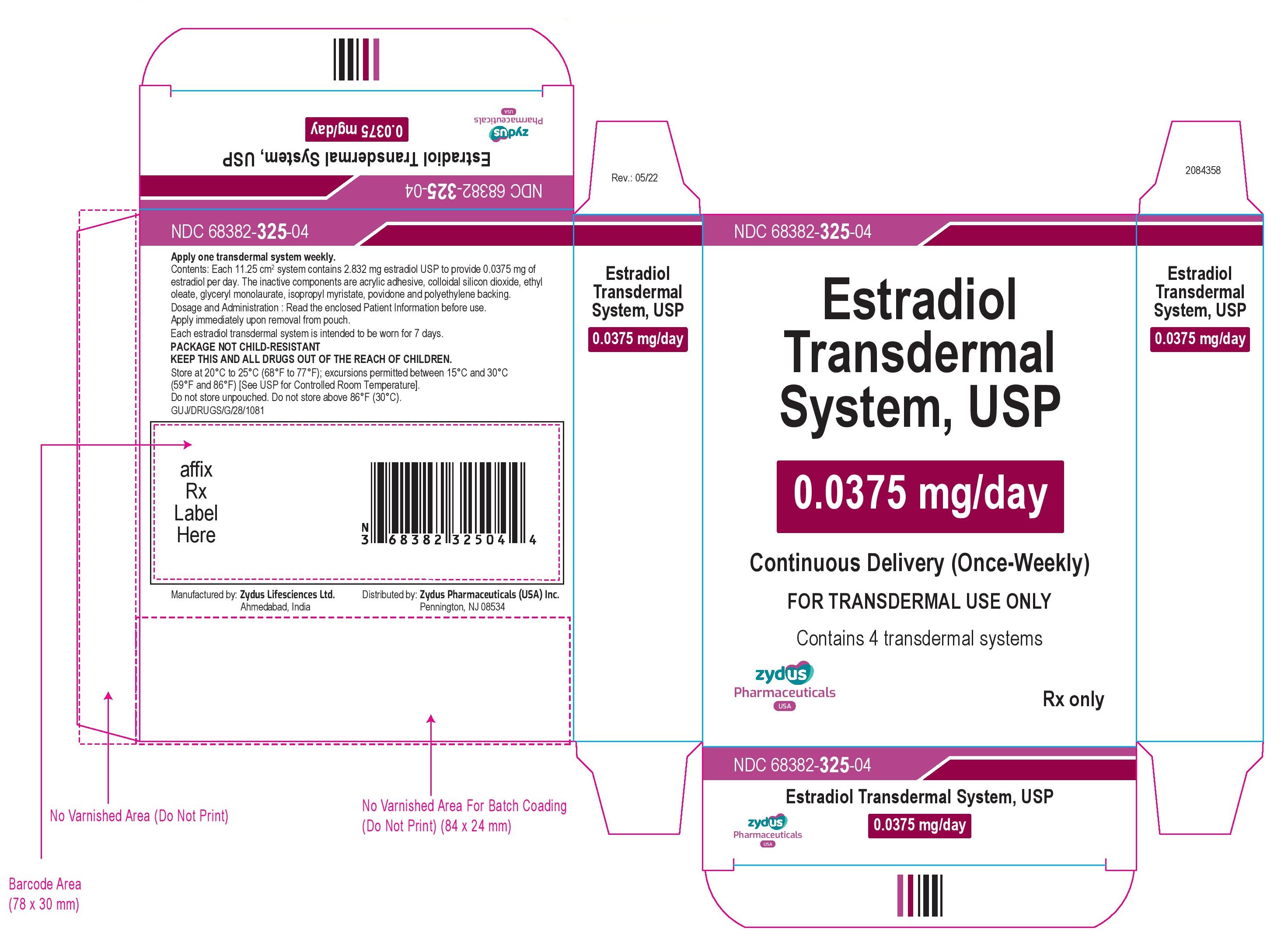
NDC 68382-325-04
Estradiol Transdermal System, USP
0.0375 mg/day
Continuous Delivery (Once-Weekly)
FOR TRANSDERMAL USE ONLY
Contains 4 transdermal systems
Rx only
zydus Pharmaceuticals USA
 Label
Label
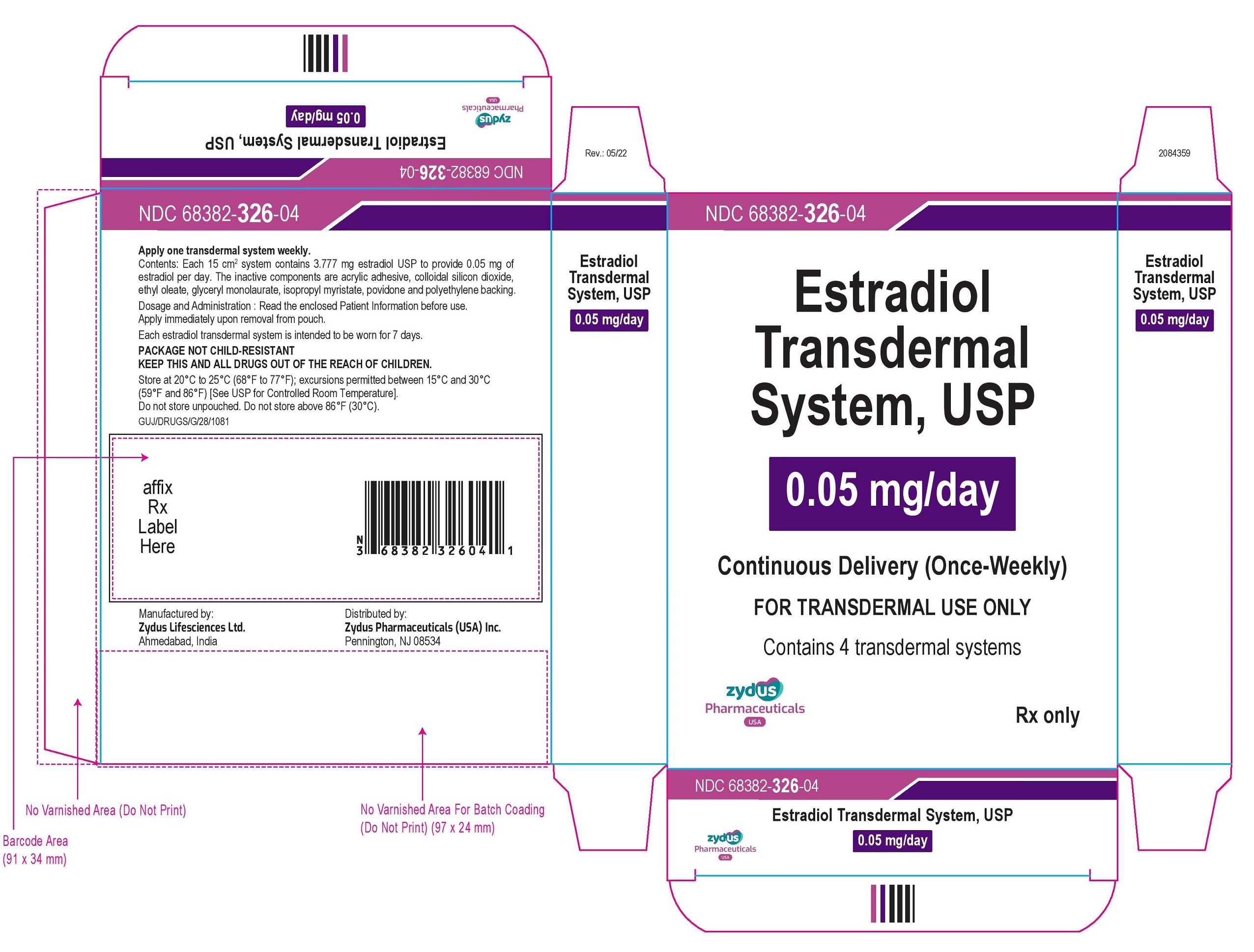
NDC 68382-326-04
Estradiol Transdermal System, USP
0.05 mg/day
Continuous Delivery (Once-Weekly)
FOR TRANSDERMAL USE ONLY
Contains 4 transdermal systems
Rx only
zydus Pharmaceuticals USA
NDC 68382-327-04
Estradiol Transdermal System, USP
0.06 mg/day
Continuous Delivery (Once-Weekly)
FOR TRANSDERMAL USE ONLY
Contains 4 transdermal systems
Rx only
zydus Pharmaceuticals USA

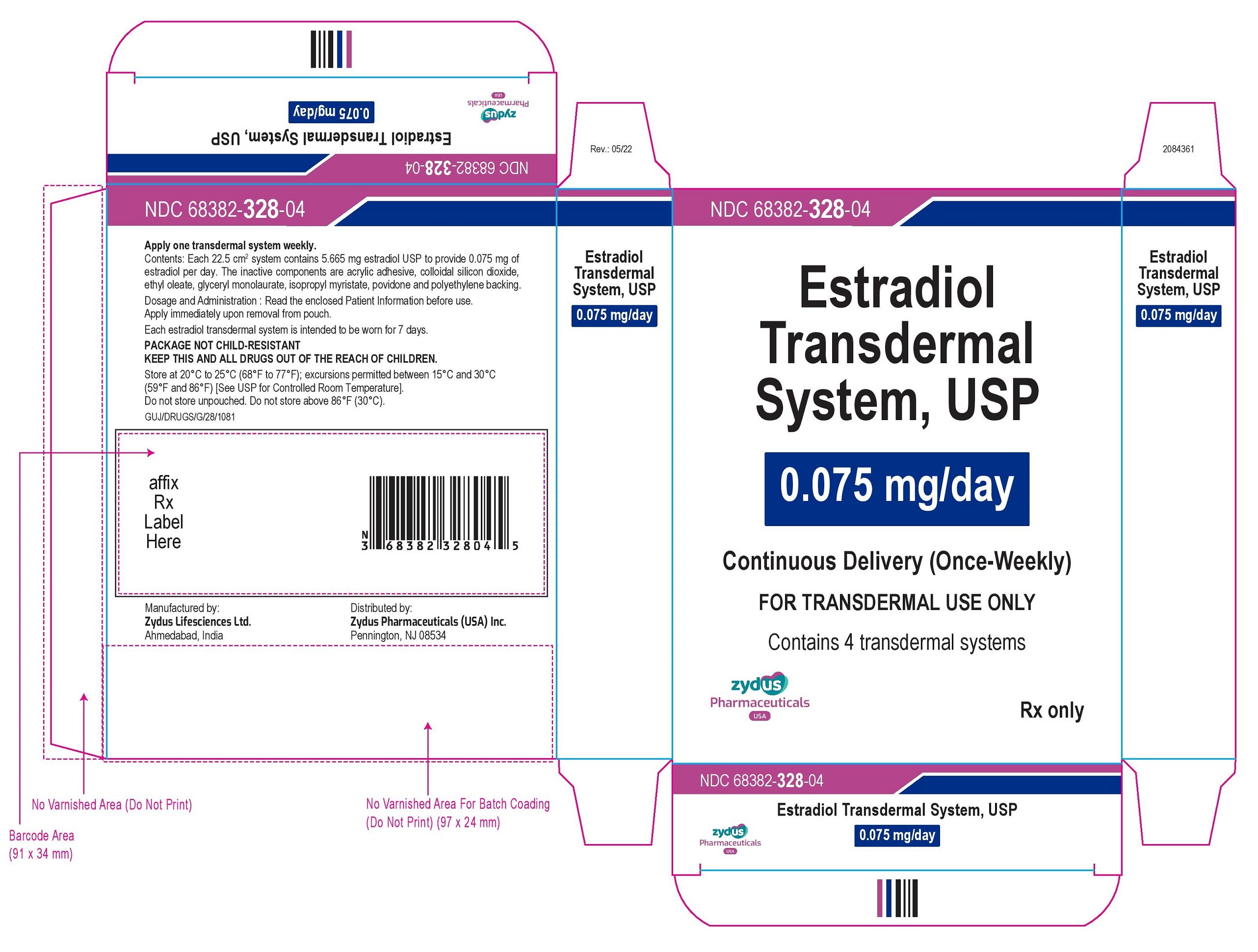
NDC 68382-328-04
Estradiol Transdermal System, USP
0.075 mg/day
Continuous Delivery (Once-Weekly)
FOR TRANSDERMAL USE ONLY
Contains 4 transdermal systems
Rx only
zydus Pharmaceuticals USA
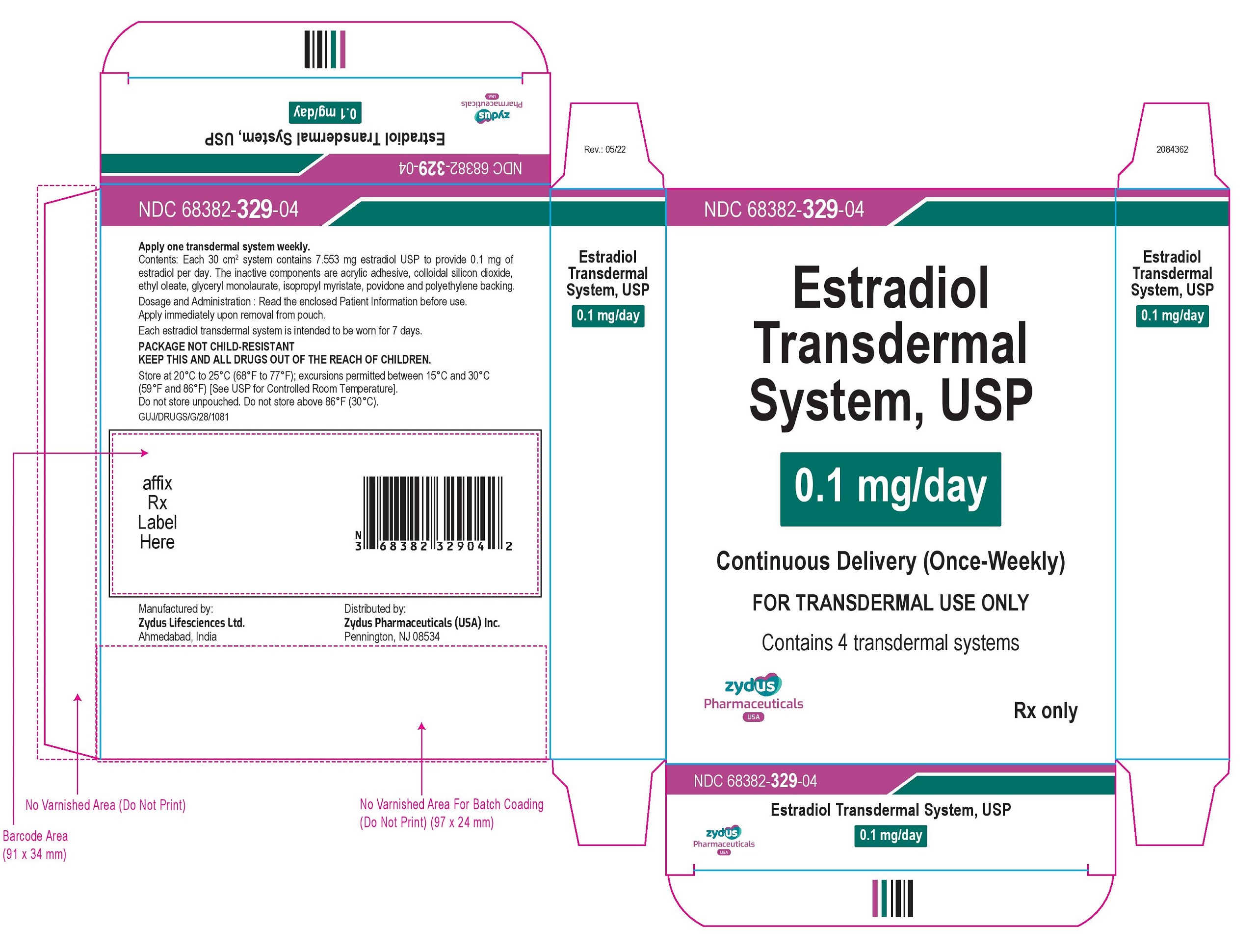
NDC 68382-329-04
Estradiol Transdermal System, USP
0.1 mg/day
Continuous Delivery (Once-Weekly)
FOR TRANSDERMAL USE ONLY
Contains 4 transdermal systems
Rx only
zydus Pharmaceuticals USA