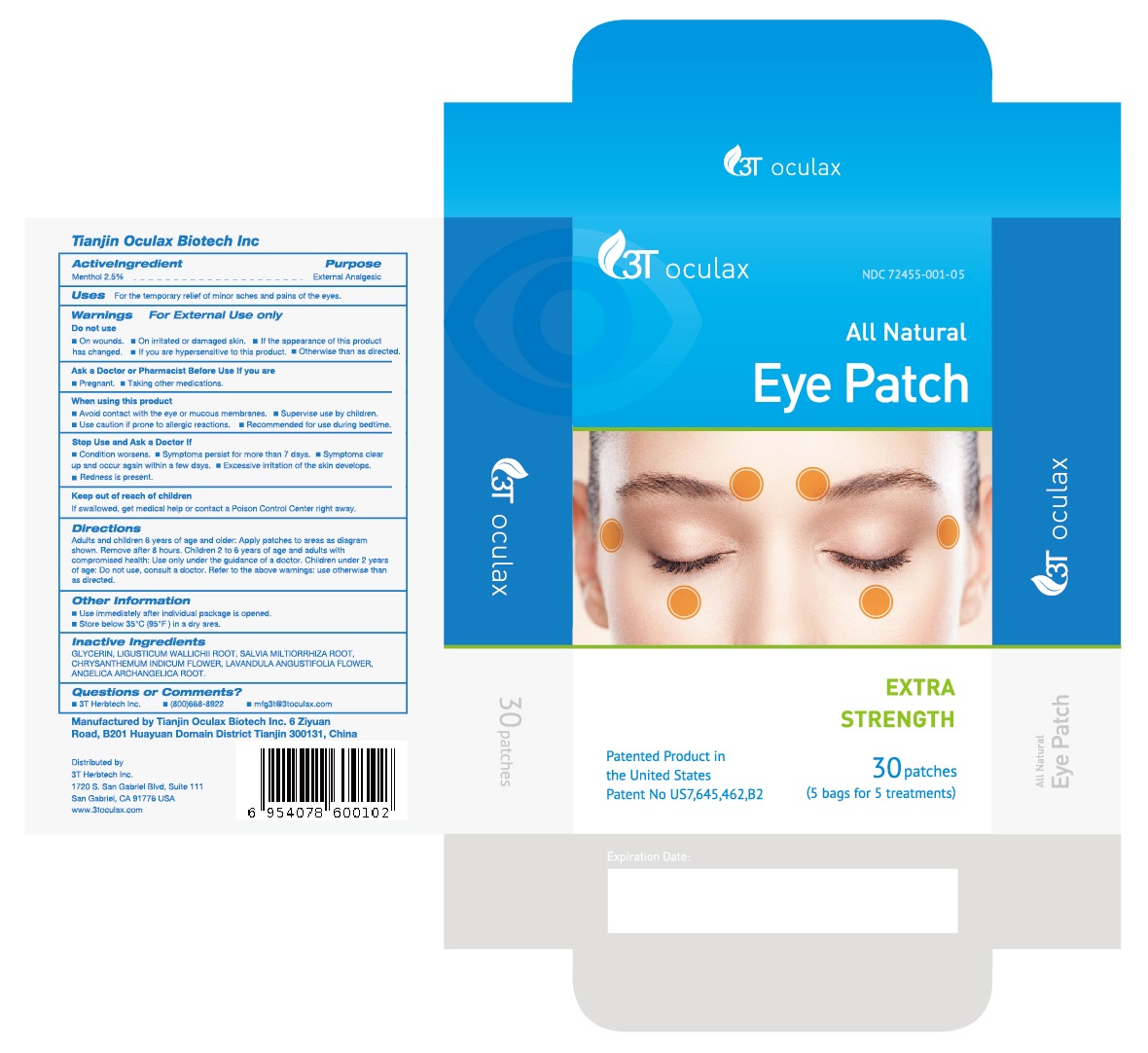
NDC Code(s) : 72455-001-05, 72455-001-10
Packager : Tianjin Oculax Biotech Inc
Category : HUMAN OTC DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| All Natural Eye PatchMenthol PATCH | |||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| LABELER - Tianjin Oculax Biotech Inc(542949666) |
| REGISTRANT - Tianjin Oculax Biotech Inc(542949666) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
| Tianjin Oculax Biotech Inc | 542949666 | manufacture(72455-001) | |
PRINCIPAL DISPLAY PANEL