NDC Code(s) : 82619-101-01, 82619-102-01
Packager : Creekwood Pharmaceuticals LLC
Category : HUMAN PRESCRIPTION DRUG LABEL
DEA Schedule : none
Marketing Status : New Drug Application
INGREDIENTS AND APPEARANCE
| FenofibrateFenofibrate TABLET | ||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| FenofibrateFenofibrate TABLET | ||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| LABELER - Creekwood Pharmaceuticals LLC(118582565) |
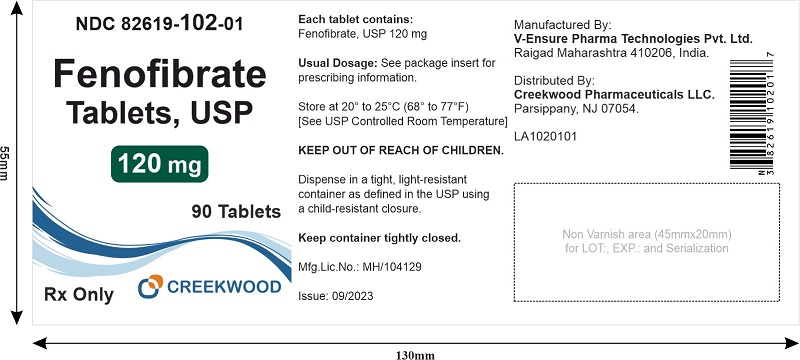
PRINCIPAL DISPLAY PANEL
Fenofibrate Tablets USP, 40 mg
NDC 82619-101-01

Fenofibrate Tablets USP, 120 mg
NDC 82619-102-01