02 Jun 2023
// CONTRACT PHARMA
17 Dec 2021
// PRESS RELEASE
26 Aug 2021
// PRESS RELEASE
Latest Content by PharmaCompass

About
Industry Trade Show
Attending
09-11 April, 2025
CPhI North America CPhI North America
Industry Trade Show
Attending
20-22 May, 2025
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
16-19 June, 2025
CONTACT DETAILS






Events
Webinars & Exhibitions
Industry Trade Show
Attending
09-11 April, 2025
CPhI North America CPhI North America
Industry Trade Show
Attending
20-22 May, 2025
BIO International Conv...BIO International Convention
Industry Trade Show
Attending
16-19 June, 2025
https://www.pharmacompass.com/speak-pharma/we-ve-quadrupled-our-production-and-added-new-customers-during-the-pandemic-speakpharma-with-wavelength-pharmaceuticals
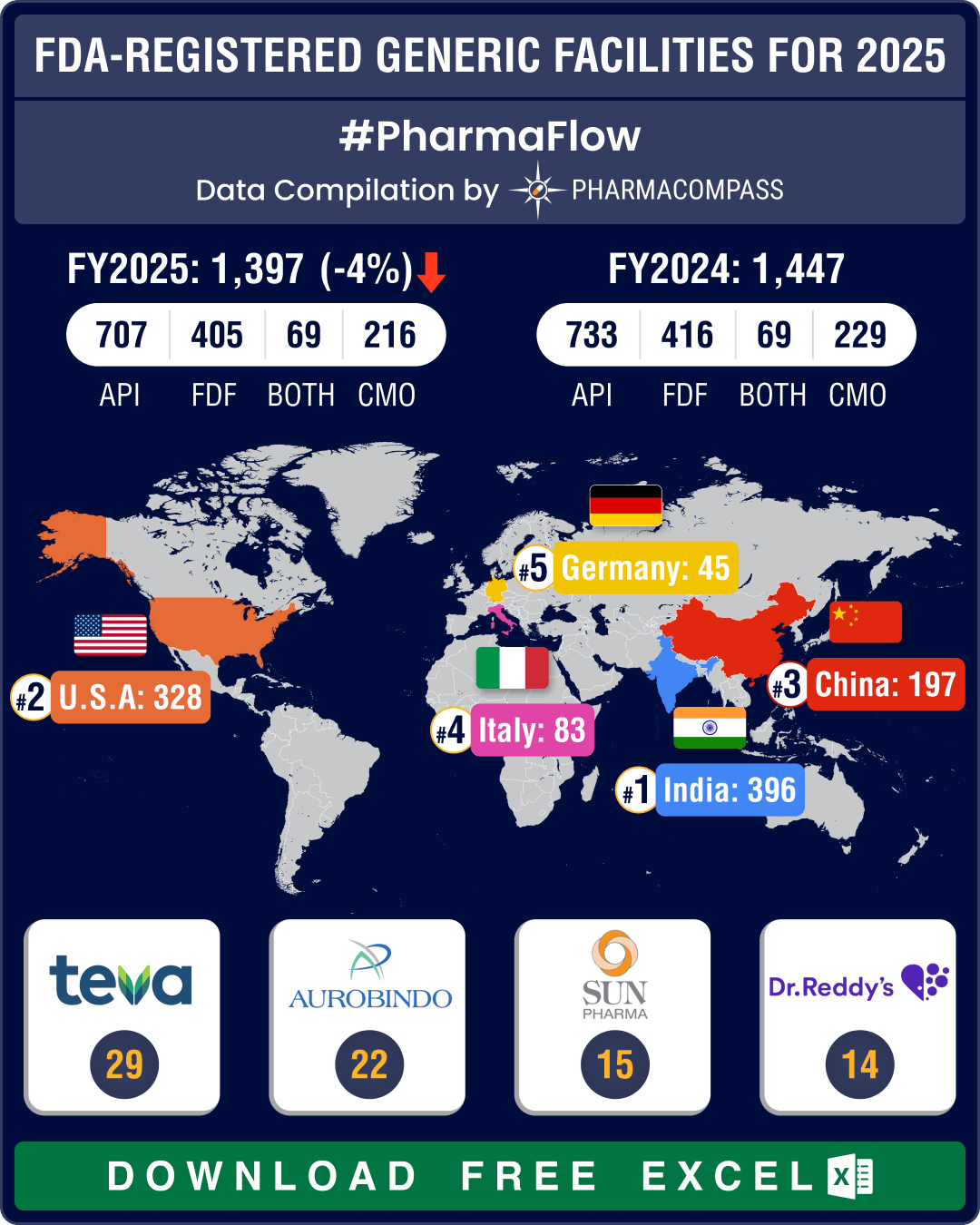
https://www.pharmacompass.com/radio-compass-blog/chinese-fda-registered-generic-facilities-gain-steam-india-maintains-lead-with-396-facilities

02 Jun 2023
// CONTRACT PHARMA
https://www.contractpharma.com/contents/view_online-exclusives/2023-02-06/ceo-spotlight-pierre-luzeau-on-seqens-us-rd-lab-investment/

17 Dec 2021
// PRESS RELEASE
https://www.wavelengthpharma.com/2021/12/17/seqens-acquisition-and-merger/

26 Aug 2021
// PRESS RELEASE
https://www.wavelengthpharma.com/2021/08/26/sk-capital-partners-to-acquire-seqens-and-merge-wavelength-pharmaceuticals/

16 Aug 2021
// PRESS RELEASE
https://www.wavelengthpharma.com/2021/08/16/wavelength-pharmaceuticals-signs-collaboration-agreement-tikun-olam-cannbit-development-manufacturing-marketing-pharma-grade-cgmp-cannabis-based-active-pharmaceutical-ingredients-api/

07 Jun 2021
// PRESS RELEASE
https://www.wavelengthpharma.com/2021/06/09/biocatalysis-an-indispensable-tool-for-api-synthesis/

19 Feb 2021
// K.Blankenship ENDPTS
https://endpts.com/israeli-api-maker-wavelength-buys-up-majority-stake-in-indian-firm-making-drug-starting-materials-and-intermediates/
Inspections and registrations
ABOUT THIS PAGE
Wavelength Pharmaceuticals is a supplier offers 3 products (APIs, Excipients or Intermediates).
Find a price of Cisatracurium Besylate bulk with DMF offered by Wavelength Pharmaceuticals
Find a price of Clascoterone bulk with DMF offered by Wavelength Pharmaceuticals
Find a price of Sitagliptin Phosphate bulk with DMF offered by Wavelength Pharmaceuticals

 Wavelength Pharmaceuticals
Wavelength Pharmaceuticals