
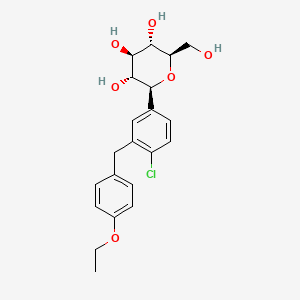

1. (2s,3r,4r,5s,6r)-2-(4-chloro-3-(4-ethoxybenzyl)phenyl)-6- (hydroxymethyl)tetrahydro-2h-pyran-3,4,5-triol
2. 2-(3-(4-ethoxybenzyl)-4-chlorophenyl)-6-hydroxymethyltetrahydro-2h-pyran-3,4,5-triol
3. Bms 512148
4. Bms-512148
5. Bms512148
6. Farxiga
7. Forxiga
1. 461432-26-8
2. Bms-512148
3. Forxiga
4. Bms 512148
5. (2s,3r,4r,5s,6r)-2-(4-chloro-3-(4-ethoxybenzyl)phenyl)-6-(hydroxymethyl)tetrahydro-2h-pyran-3,4,5-triol
6. Dagagflozin
7. (1s)-1,5-anhydro-1-c-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-d-glucitol
8. 1ull0qj8uc
9. Chembl429910
10. Chebi:85078
11. Lyn-045
12. Dapagliflozin [usan:inn]
13. (2s,3r,4r,5s,6r)-2-(4-chloro-3-(4-ethoxybenzyl)phenyl)-6- (hydroxymethyl)tetrahydro-2h-pyran-3,4,5-triol
14. (2s,3r,4r,5s,6r)-2-{4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl}-6-(hydroxymethyl)oxane-3,4,5-triol
15. Bms512148
16. Forxiga (tn)
17. Chembl3125458
18. D-glucitol, 1,5-anhydro-1-c-(4-chloro-3-((4-ethoxyphenyl)methyl)phenyl)-, (1s)-
19. (2s,3r,4r,5s,6r)-2-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-6-(hydroxymethyl)oxane-3,4,5-triol
20. Dapagliflozin [usan]
21. Unii-1ull0qj8uc
22. Bms5121458
23. S1548_selleck
24. C-aryl Glucoside, 6
25. Dapagliflozin [mi]
26. Dapagliflozin [inn]
27. Dapagliflozin (usan/inn)
28. 2-(3-(4-ethoxybenzyl)-4-chlorophenyl)-6-hydroxymethyltetrahydro-2h-pyran-3,4,5-triol
29. Dapagliflozin [vandf]
30. Schembl157820
31. Dapagliflozin [who-dd]
32. Gtpl4594
33. Bdbm20880
34. Dtxsid20905104
35. Ex-a005
36. Bcpp000265
37. Dapagliflozin [orange Book]
38. Amy18541
39. Zinc3819138
40. Bdbm50448923
41. Mfcd13182359
42. S1548
43. Qternmet Component Dapaglifozin
44. Akos005145763
45. Dapagliflozin Component Qternmet
46. Bcp9000583
47. Bl-0052
48. Ccg-229917
49. Cs-0781
50. Db06292
51. Ex-7214
52. Ncgc00250402-09
53. Ac-24699
54. Bd164346
55. Hy-10450
56. Qternmet Xr Component Dapagliflozin
57. Dapagliflozin Component Of Qternmet Xr
58. A25150
59. C22193
60. D08897
61. 432d268
62. Q409898
63. J-500392
64. Brd-k58160573-001-01-3
65. Brd-k58160573-001-05-4
66. 1-[3-(beta-d-glucopyranosyl)-6-chlorobenzyl]-4-ethoxybenzene
67. (1s)-1,5-anhydro-1-[4-chloro-3-(4-ethoxybenzyl)phenyl]-d-glucitol
68. (2s, 3r, 4r, 5s, 6r)-2-[4-chloro-3-(4-ethoxy-benzyl)-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol
69. (2s,3r,4r,5s,6r)-2-(3-(4-ethoxybenzyl)-4-chlorophenyl)-6-(hydroxymethyl)-tetrahydro-2h-pyran-3,4,5-triol
70. (2s,3r,4r,5s,6r)-2-(3-(4-ethoxybenzyl)-4-chlorophenyl)-6-(hydroxymethyl)-tetrahydro-2h-pyran-3,4,5-triol;bms-512148
71. (2s,3r,4r,5s,6r)-2-[4-chloro-3-(4-ethoxy-benzyl)-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol
| Molecular Weight | 408.9 g/mol |
|---|---|
| Molecular Formula | C21H25ClO6 |
| XLogP3 | 2.3 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Exact Mass | 408.1339662 g/mol |
| Monoisotopic Mass | 408.1339662 g/mol |
| Topological Polar Surface Area | 99.4 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 472 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 3 | |
|---|---|
| Drug Name | FARXIGA |
| Active Ingredient | DAPAGLIFLOZIN |
| Company | ASTRAZENECA AB (Application Number: N202293. Patents: 6414126, 6515117, 6936590, 7456254, 7851502, 7919598, 8221786, 8329648, 8361972, 8431685, 8461105, 8501698, 8685934, 8716251, 8721615, 8906851, 9198925, 9238076) |
| 2 of 3 | |
|---|---|
| Drug Name | XIGDUO XR |
| Active Ingredient | DAPAGLIFLOZIN; METFORMIN HYDROCHLORIDE |
| Company | ASTRAZENECA AB (Application Number: N205649. Patents: 6414126, 6515117, 6936590, 7919598, 8501698, 8685934, 9198925, 9616028) |
| 3 of 3 | |
|---|---|
| Drug Name | QTERN |
| Active Ingredient | DAPAGLIFLOZIN; SAXAGLIPTIN HYDROCHLORIDE |
| Company | ASTRAZENECA AB (Application Number: N209091. Patents: 6414126, 6515117, 6936590, 7919598, 8221786, 8361972, 8501698, 8628799, 8716251, 9198925, RE44186) |
Dapagliflozin is indicated to improve glycemic control in adult patients with type 2 diabetes mellitus along with diet and exercise.
FDA Label
Forxiga is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise
- as monotherapy when metformin is considered inappropriate due to intolerance.
- in addition to other medicinal products for the treatment of type 2 diabetes.
For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied.
Heart failure
- Forxiga is indicated in adults for the treatment of symptomatic chronic heart failure with reduced ejection fraction.
Chronic kidney disease
- Forxiga is indicated in adults for the treatment of chronic kidney disease.
Type 2 diabetes mellitus
Edistride is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise
- as monotherapy when metformin is considered inappropriate due to intolerance.
- in addition to other medicinal products for the treatment of type 2 diabetes.
For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4. 4, 4. 5 and 5. 1.
Heart failure
Edistride is indicated in adults for the treatment of symptomatic chronic heart failure with reduced ejection fraction.
Chronic kidney disease
Edistride is indicated in adults for the treatment of chronic kidney disease.
Treatment of chronic kidney disease
Prevention of cardiovascular events in patients with chronic heart failure
Treatment of type I diabetes mellitus
Treatment of type II diabetes mellitus
Treatment of ischaemic heart disease
Treatment of Coronavirus disease 2019 (COVID-19)
Dapagliflozin inhibits the sodium-glucose contransporter 2(SGLT2) which is primarily located in the proximal tubule of the nephron. SGLT2 facilitates 90% of glucose resorption in the kidneys and so its inhibition allows for glucose to be excreted in the urine. This excretion allows for better glycemic control and potentially weight loss in patients with type 2 diabetes mellitus.
Sodium-Glucose Transporter 2 Inhibitors
Compounds that inhibit SODIUM-GLUCOSE TRANSPORTER 2. They lower blood sugar by preventing the reabsorption of glucose by the kidney and are used in the treatment of TYPE 2 DIABETES MELLITUS. (See all compounds classified as Sodium-Glucose Transporter 2 Inhibitors.)
A10BK01
A10BK01
A10BK01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
A - Alimentary tract and metabolism
A10 - Drugs used in diabetes
A10B - Blood glucose lowering drugs, excl. insulins
A10BK - Sodium-glucose co-transporter 2 (sglt2) inhibitors
A10BK01 - Dapagliflozin
Absorption
Oral dapagliflozin reaches a maximum concentration within 1 hour of administration when patients have been fasting. When patients have consumed a high fat meal, the time to maximum concentration increases to 2 hours and the maximum concentration decreases by half though a dose adjustment is not necessary. Oral dapagliflozin is 78% bioavailable.
Route of Elimination
75.2% of dapagliflozin is recovered in the urine with 1.6% of the dose unchanged by metabolism. 21% of the dose is excreted in the feces with 15% of the dose unchanged by metabolism.
Volume of Distribution
118L.
Clearance
Oral plasma clearance was 4.9 mL/min/kg, and renal clearance was 5.6 mL/min.
Dapagliflozin is primarily glucuronidated to become the inactive 3-O-glucuronide metabolite(60.7%). Dapagliflozin also produces another minor glucuronidated metabolite(5.4%), a de-ethylated metabolite(<5%), and a hydroxylated metabolite(<5%). Metabolism of dapagliflozin is mediated by cytochrome p-450(CYP)1A1, CYP1A2, CYP2A6, CYP2C9, CYP2D6, CYP3A4, uridine diphosphate glucuronyltransferase(UGT)1A9, UGT2B4, and UGT2B7. Glucuronidation to the major metabolite is mediated by UGT1A9.
13.8h.
Dapagliflozin inhibits the sodium-glucose contransporter 2(SGLT2) which is primarily located in the proximal tubule of the nephron. SGLT2 facilitates 90% of glucose resorption in the kidneys and so its inhibition allows for glucose to be excreted in the urine. This excretion allows for better glycemic control and potentially weight loss in patients with type 2 diabetes mellitus.
BUILDING BLOCK


