By PharmaCompass
2024-06-06
Impressions: 2613
Pharma indices have rebounded after ending March and April in the red. May saw the Nasdaq Biotechnology Index (NBI) spike 5.6 percent from 4,168.63 to 4,401.1 and the SPDR S&P Biotech ETF (XBI) index rise 5.1 percent from 84.82 to 89.13. The S&P Biotechnology Select Industry Index (SPSIBI) saw a 5.3 percent gain to 6,930.67 from 6,584.4 at the end of April.
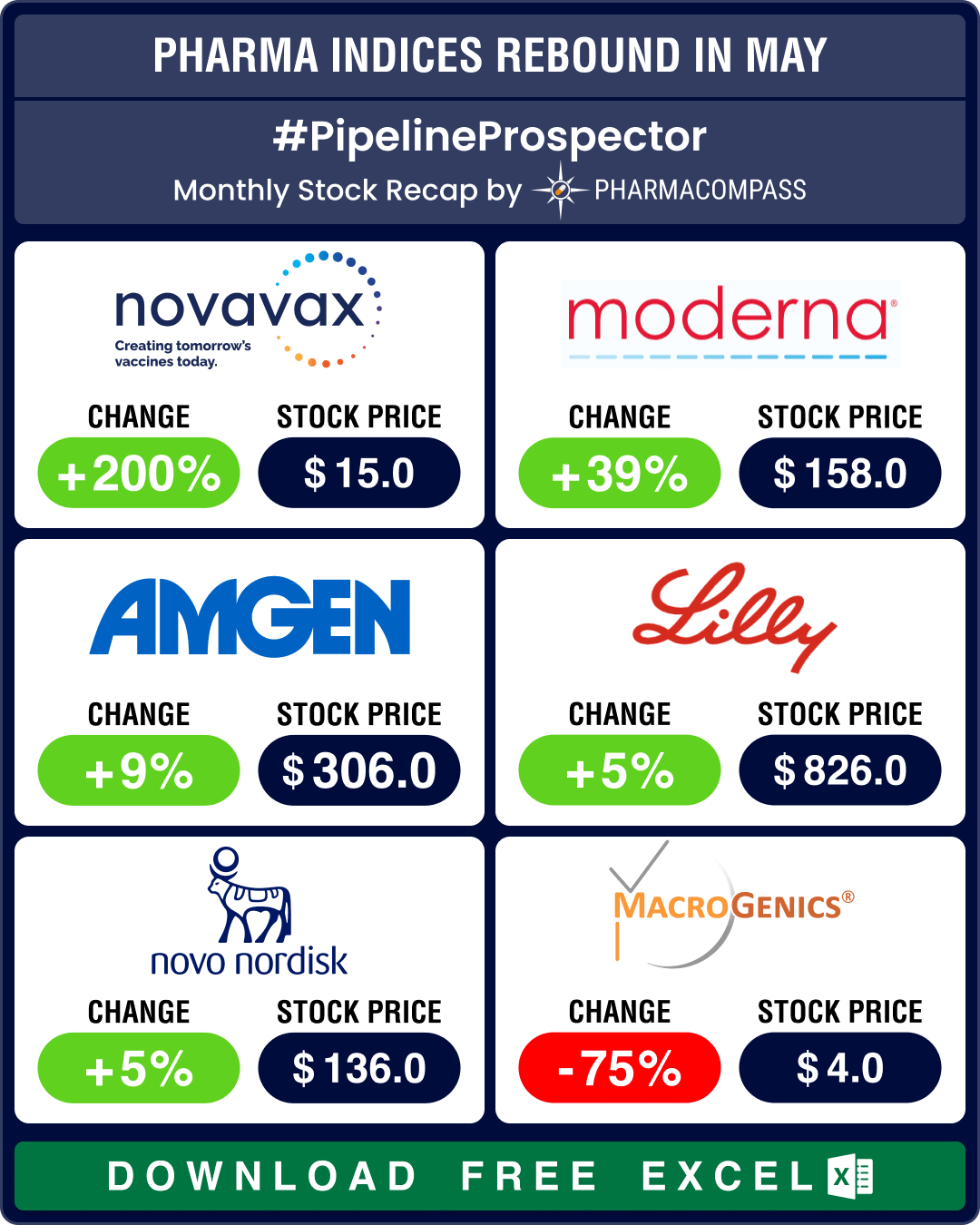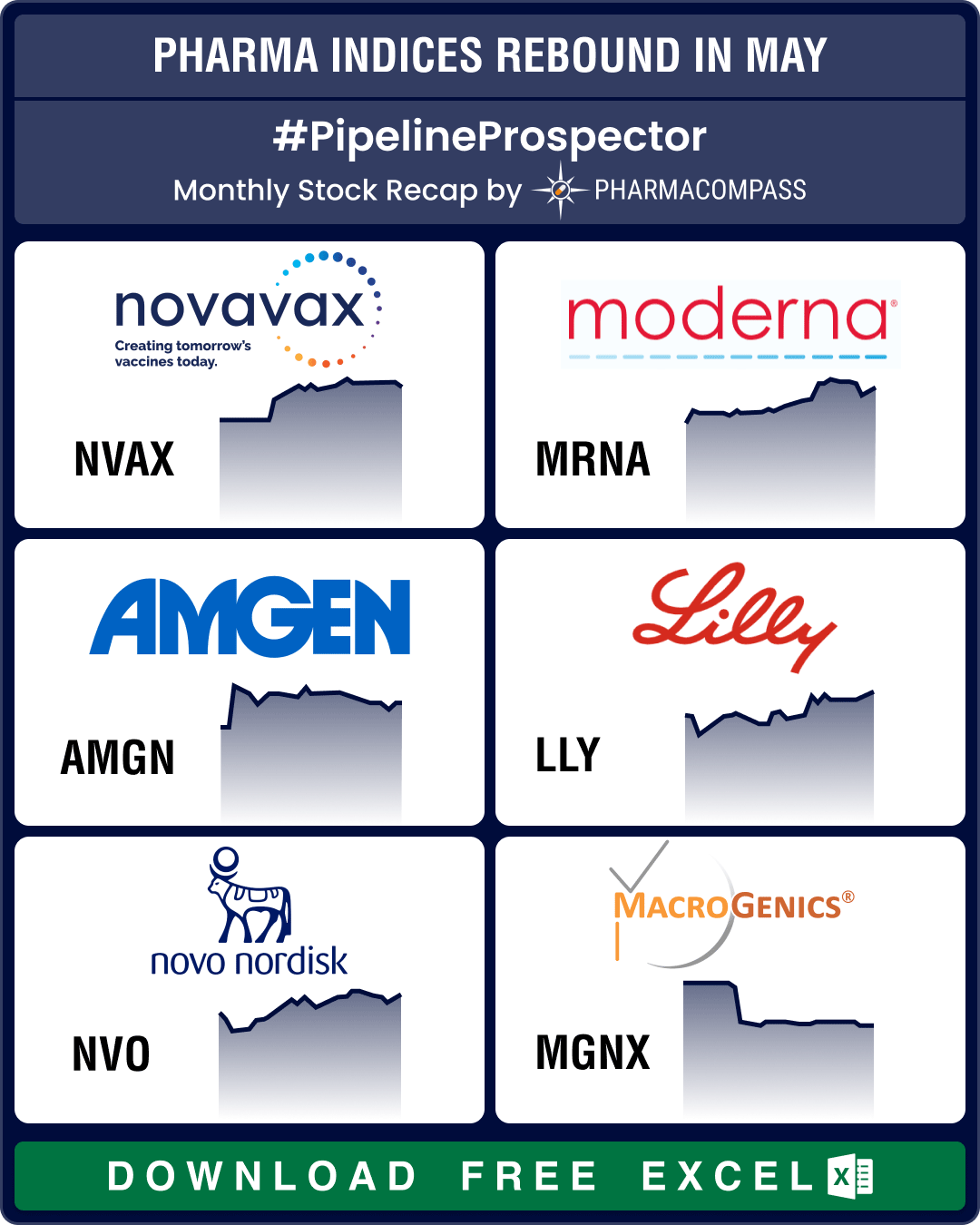
The month didn’t see very many approvals from the US Food and Drug Administration (FDA). But what May lacked in approvals, it made up in acquisitions, deals, and trial news. Among the noteworthy approvals were Amgen’s (stock up 9 percent) Imdelltra, which won FDA’s accelerated approval to treat adults in the advanced stages of small cell lung cancer (SCLC). The agency okayed Tris Pharma’s Onyda XR, a once-a-day treatment for attention deficit hyperactivity disorder (ADHD) for patients six years and older. This makes it the first liquid non-stimulant ADHD drug approved in the US.
Bristol Myers Squibb’s Breyanzi saw two FDA approvals in May. The agency first expanded its use to treat adults with a type of blood cancer known as follicular lymphoma. FDA then okayed it to treat relapsed or refractory mantle cell lymphoma. With five approvals, Breyanzi is the only CAR T cell therapy approved in the US for four distinct subtypes of non-Hodgkin lymphoma, bringing this personalized therapy to the broadest array of patients with B-cell malignancies. And FDA approved Moderna’s (stock up 39 percent) mRESVIA, an mRNA respiratory syncytial virus (RSV) vaccine, to protect adults aged 60 years and older from lower respiratory tract disease caused by RSV infection.
Access the Pipeline Prospector Dashboard for May 2024 Newsmakers (Free Excel)
J&J pays US$ 2.1 bn for eczema drug assets; Merck to buy EyeBio for up to US$ 3 bn
Seeking to address the significant need in atopic dermatitis (eczema), J&J spent US$ 2.1 billion on bispecific antibody candidates. This included acquiring Proteologix for US$ 850 million and rights to Numab Therapeutics' NM26 for US$ 1.25 billion. NM26 is a promising phase 2-ready treatment.
Bolstering its ophthalmology portfolio, Merck is acquiring EyeBio for up to US$ 3 billion. The deal includes an upfront payment of US$ 1.3 billion and grants Merck access to EyeBio's lead candidate, Restoret, for diabetic macular edema and neovascular age-related macular degeneration.
In a US$ 1.8 billion deal (including US$ 1.15 billion upfront), Biogen acquired Human Immunology Biosciences (HI-Bio) to gain access to felzartamab, a potential cure for various immune-mediated diseases. Expanding its oncology portfolio, Novartis acquired Mariana Oncology for US$ 1 billion upfront and US $750 million in milestone payments. Mariana focuses on developing novel radioligand therapies to treat cancers with high unmet needs. Aiming to become a major player in the pharmaceutical industry, Japanese conglomerate Asahi Kasei offered US$ 1.1 billion to acquire Swedish drugmaker Calliditas Therapeutics.
Access the Pipeline Prospector Dashboard for May 2024 Newsmakers (Free Excel)
Sanofi bails out Novavax with US$ 1.2 bn deal; Takeda nabs option to license Alzheimer’s therapies
Novavax has received a timely shot in the arm, as Sanofi will pay the former up to US$ 1.2 billion in exchange for a license to commercialize its existing Covid vaccine and develop a combo jab to protect against Covid and influenza. The struggling Maryland biotech’s management, or lack thereof, had come under fire from investors. Sanofi also gets a minority 4.9 percent stake in Novavax. Following this, Novavax raised its 2024 revenue guidance from between US$ 800 million and US$ 1 billion to between US$ 970 million and US$ 1.17 billion. Novavax’s stock rocketed 200 percent in May.
Japanese pharmaceutical giant Takeda made two significant moves. After an upfront payment of US$ 100 million, Takeda now has the exclusive option to secure global rights to AC Immune's Alzheimer's vaccine and immunotherapies targeting amyloid beta for another US$ 2.1 billion. Additionally, Takeda partnered with Shanghai-based Degron Therapeutics in a deal valued at up to US$1.2 billion. This collaboration focuses on discovering and developing novel cancer, neurological, and inflammatory drugs using a promising new approach called "molecular glue degraders."
Access the Pipeline Prospector Dashboard for May 2024 Newsmakers (Free Excel)
Novo scores late-stage wins in hemophilia, kidney disease; Lilly’s weekly insulin proves to be as effective as daily doses
Novo Nordisk’s Mim8 has hit its primary endpoint in a late-stage trial to treat people with hemophilia A. This paves the way for filing for approval later this year and competing with Roche’s blockbuster Hemlibra. Meanwhile, Ozempic lowered the risk of death in type 2 diabetes patients with chronic kidney disease. A late-stage trial with 3,533 people showed weekly semaglutide injections reduced kidney failure and death due to kidney complications by 24 percent.
However, Novo did suffer a setback this month. FDA’s advisory panel voted seven to four that the benefits of Novo’s once-weekly insulin, Awiqli, do not outweigh its risks. The Danish drugmaker is in a race with Lilly to bring to market weekly injections with long-acting insulins, which would lower the treatment burden for diabetes patients. An increased risk of low blood sugar was flagged by the advisors in patients with type 1 diabetes. Novo's sales increased 24 percent to DKK 65.3 billion (US$ 9.4 billion) in the first quarter of 2024.
Eli Lilly’s once-weekly insulin jab, efsitora, showed blood sugar reduction consistent with the daily insulins widely used today by patients with type 2 diabetes. Efsitora met its primary endpoints in two phase 3 trials. The Indiana drugmaker said it is investing an additional US$ 5.3 billion at its Lebanon (Indiana, US) manufacturing site to boost the supply of Zepbound and Mounjaro.
Access the Pipeline Prospector Dashboard for May 2024 Newsmakers (Free Excel)
Novartis tees up two rare kidney disease therapy approvals; Bayer’s menopause drug reduces hot flashes
Novartis touted two promising late-stage readouts for rare kidney disease treatments as it tees up two potential approvals in the renal therapy space. The Swiss drugmaker’s experimental drug, atrasentan, reduced proteinuria (elevated protein in the urine) by 36.1 percent in IgA nephropathy patients. Fabhalta achieved a 35.1 percent proteinuria reduction in patients with the ultra-rare kidney disease C3 glomerulopathy (C3G). C3G currently has no approved therapies.Bayer said its non-hormonal drug, elinzanetant, significantly reduced debilitating bursts of body heat (hot flashes) associated with menopause. This tees it up to become only the second non-hormonal drug to do so after Astellas’ Veozah.
Access the Pipeline Prospector Dashboard for May 2024 Newsmakers (Free Excel)
Our view
For long, mental health researchers have been captivated by psychoactive ingredients found in cannabis and LSD. However, advocates suffered a setback in early June when an FDA advisory panel voted against the first MDMA treatment for post-traumatic stress disorder (PTSD). Still, June kicked off with the American Society of Clinical Oncology (ASCO) annual meeting in Chicago, which saw tons of developments in treating cancers. It is shaping up to be an interesting month with over a dozen Prescription Drug User Fee Act (PDUFA) dates including for two drugs to treat chronic obstructive pulmonary disease (COPD) – Dupixent and ensifentrine.
Access the Pipeline Prospector Dashboard for May 2024 Newsmakers (Free Excel)
Pharma & Biotech Newsmakers in May 2024
| Company | Country | Currency | Market Cap (Bn) | Change In Market Cap (M) | Stock Price | Change In Price |
|---|
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
Image Credit : Pharma & Biotech Newsmakers in May 2024 by PharmaCompass license under CC BY 2.0
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”








