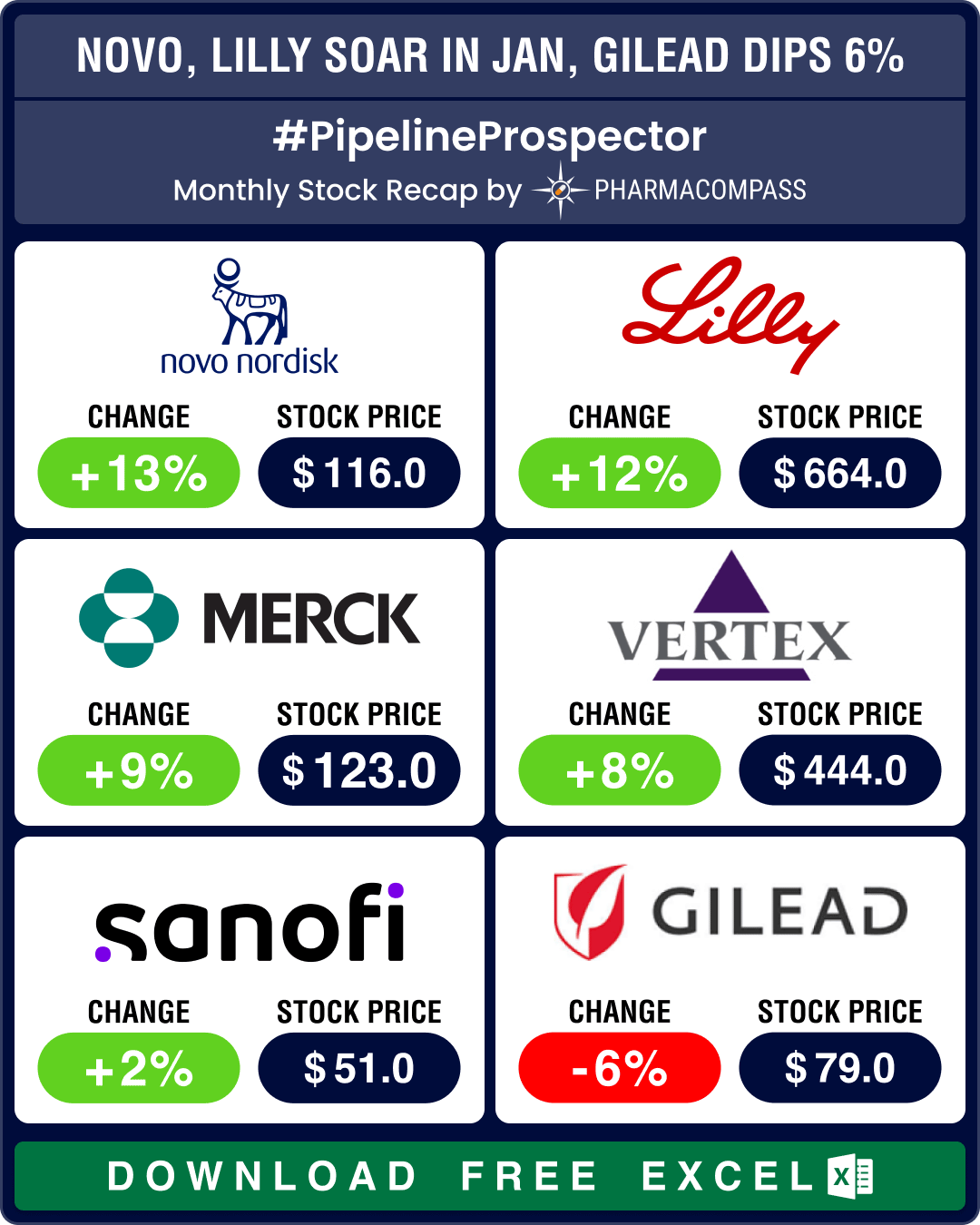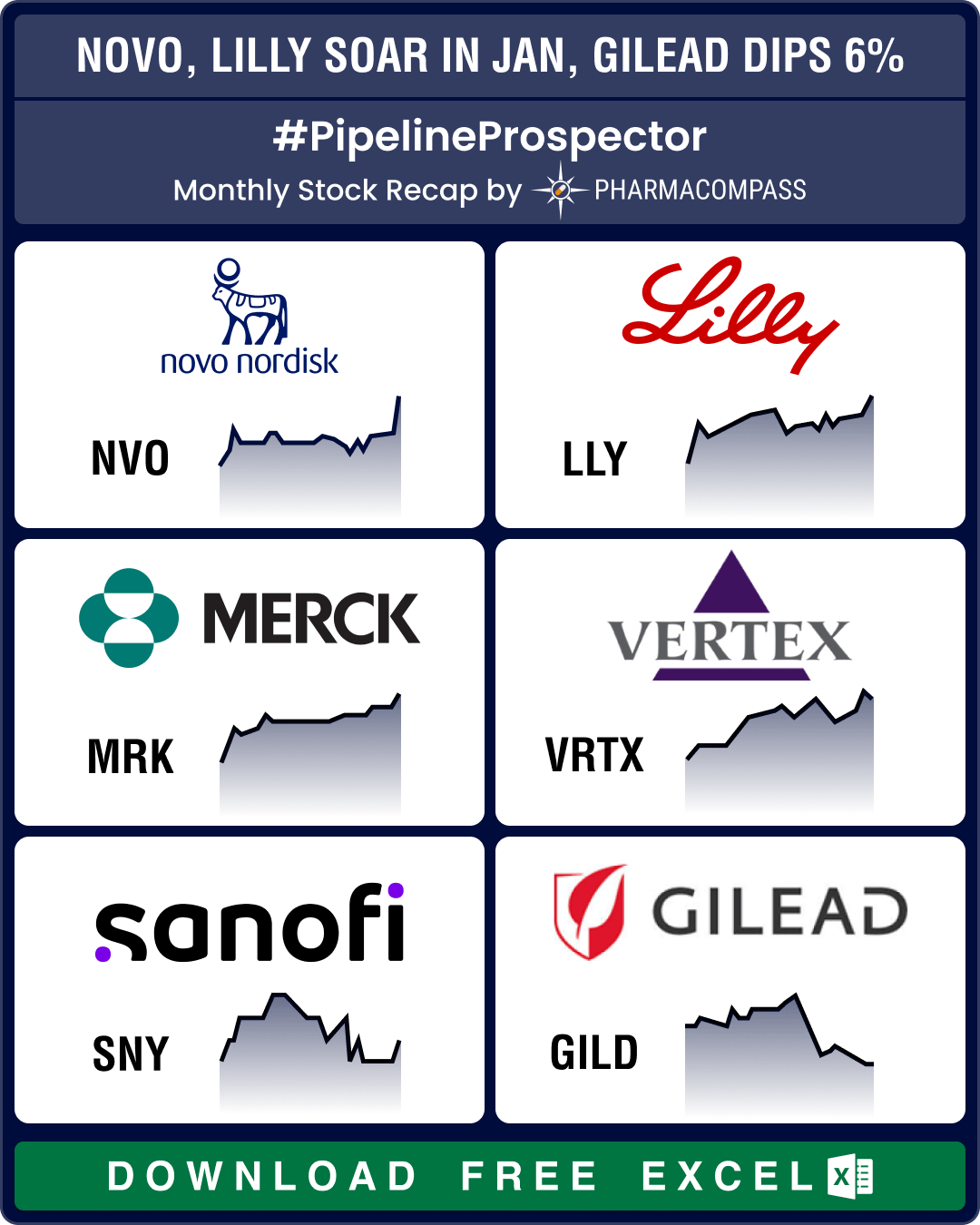
Stock Recap #PipelineProspector
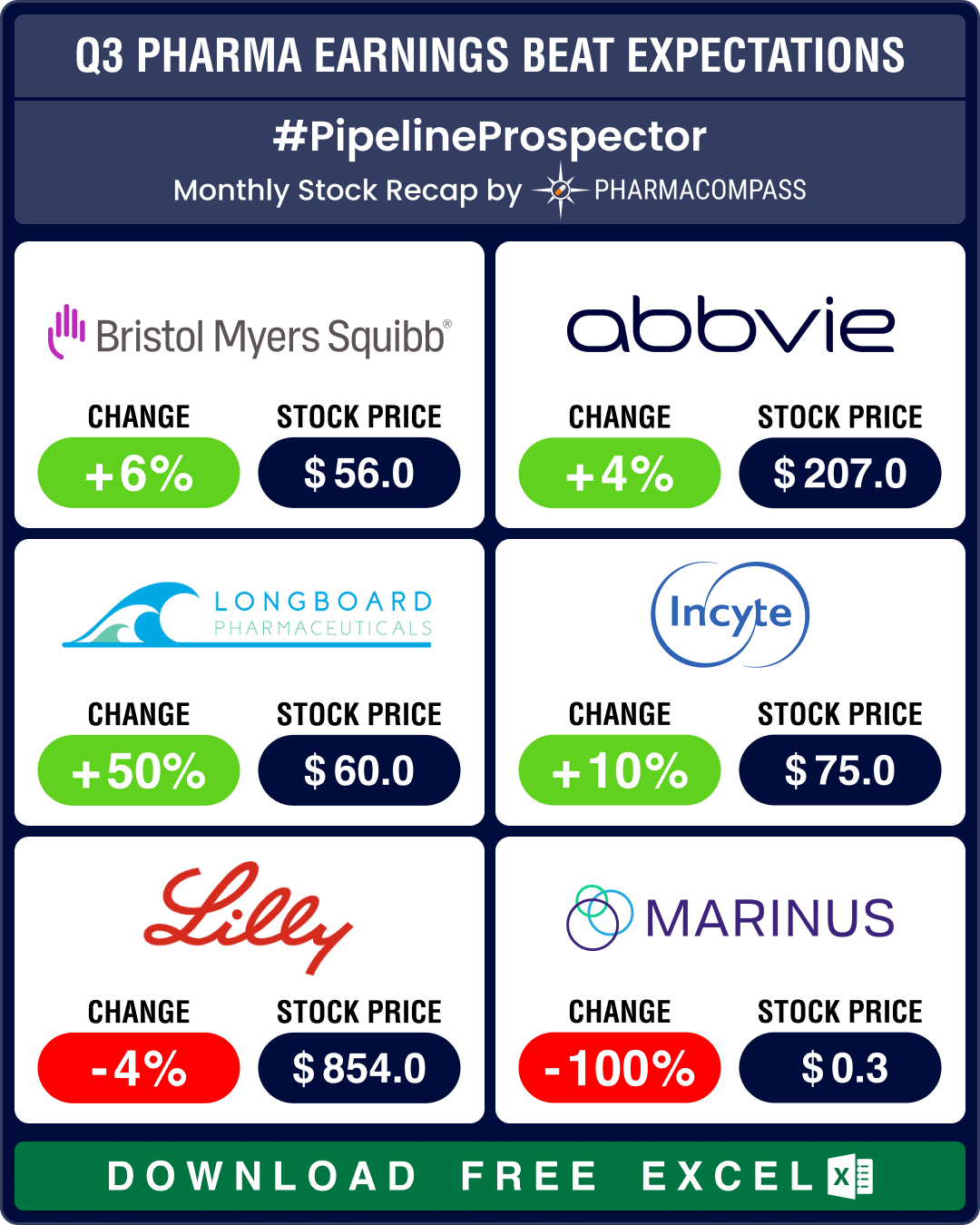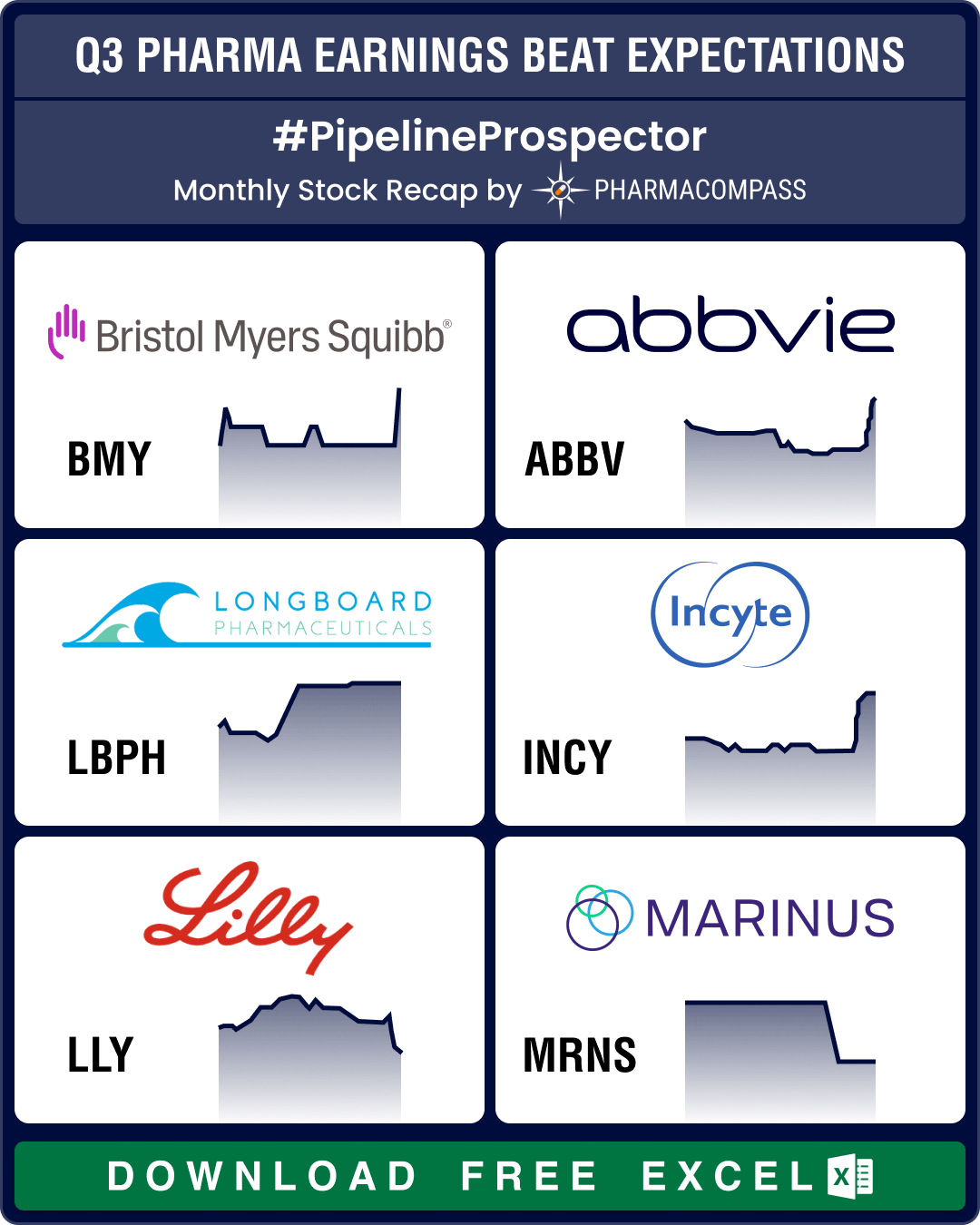
Pipeline Prospector Oct 2024: Lundbeck acquires Longboard for US$ 2.6 bn; molecular glue degrader tech witnesses dealmaking
In October, several pharma companies posted their third quarter (Q3) results. Drugmakers like Pfizer
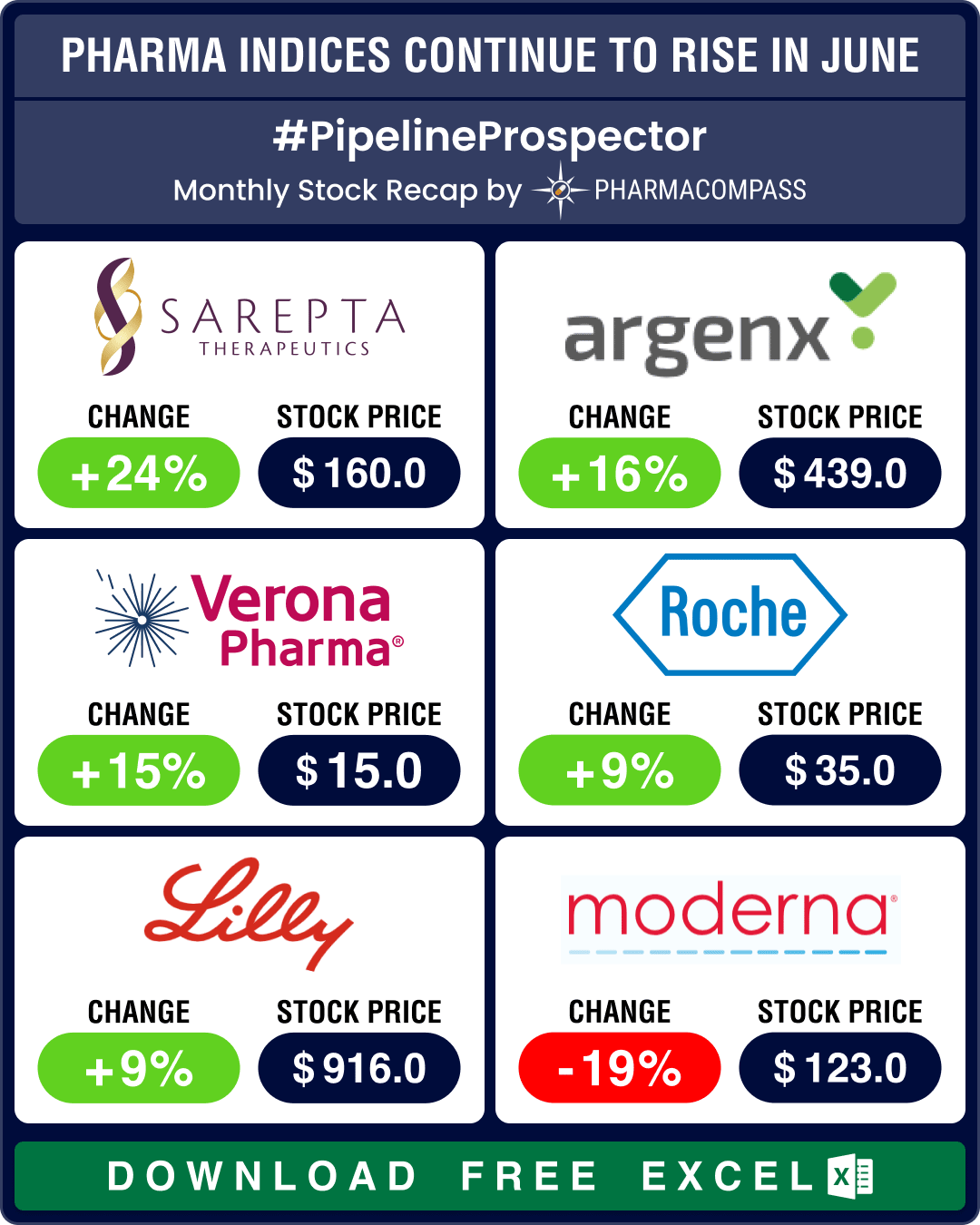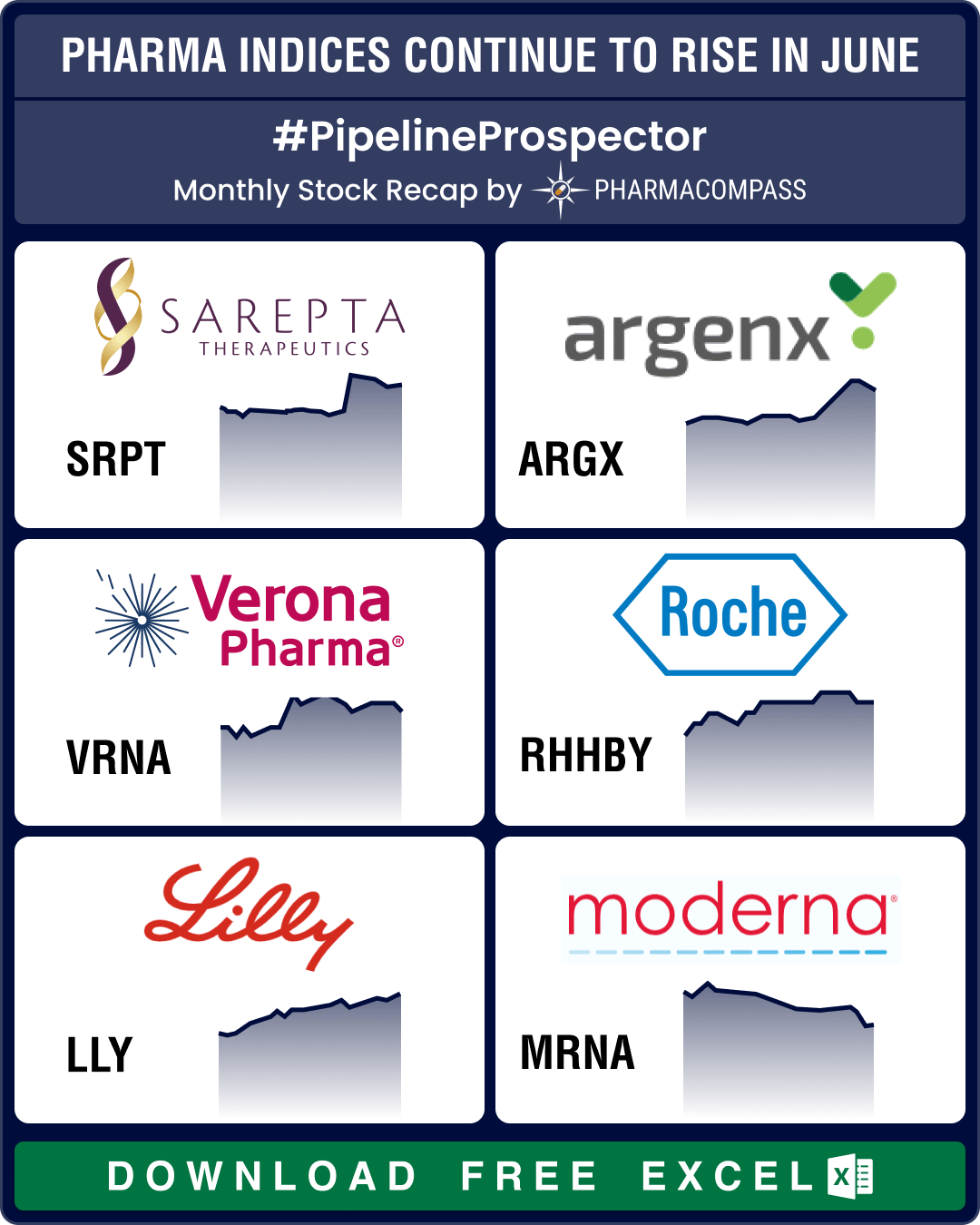
Pipeline Prospector June 2024: FDA approves Merck’s next-gen pneumococcal vaccine, Verona’s COPD therapy
The pharma indices were back in the black in May, and the good streak continued through June with th


 Market Place
Market Place Sourcing Support
Sourcing Support