By PharmaCompass
2025-04-03
Impressions: 4200
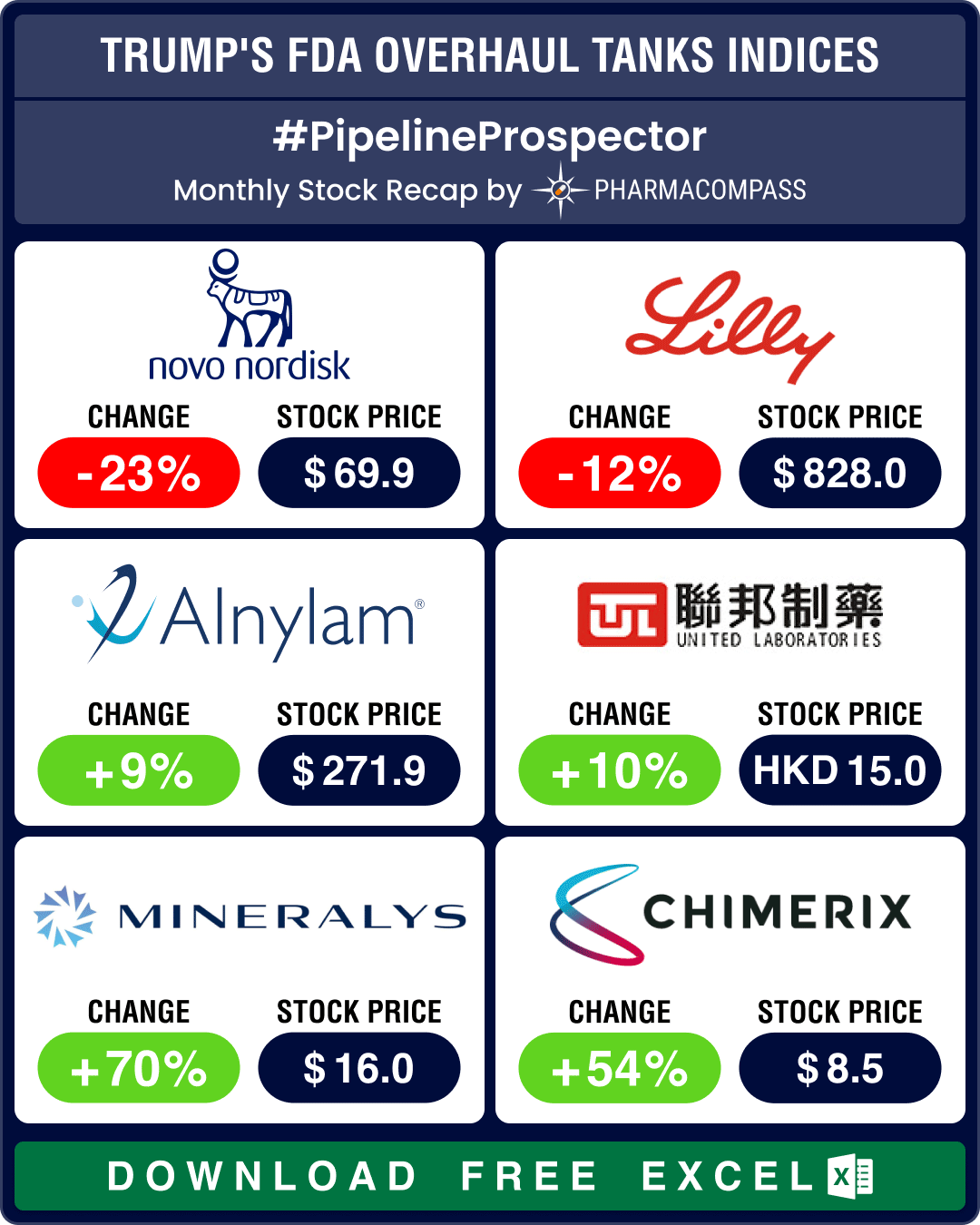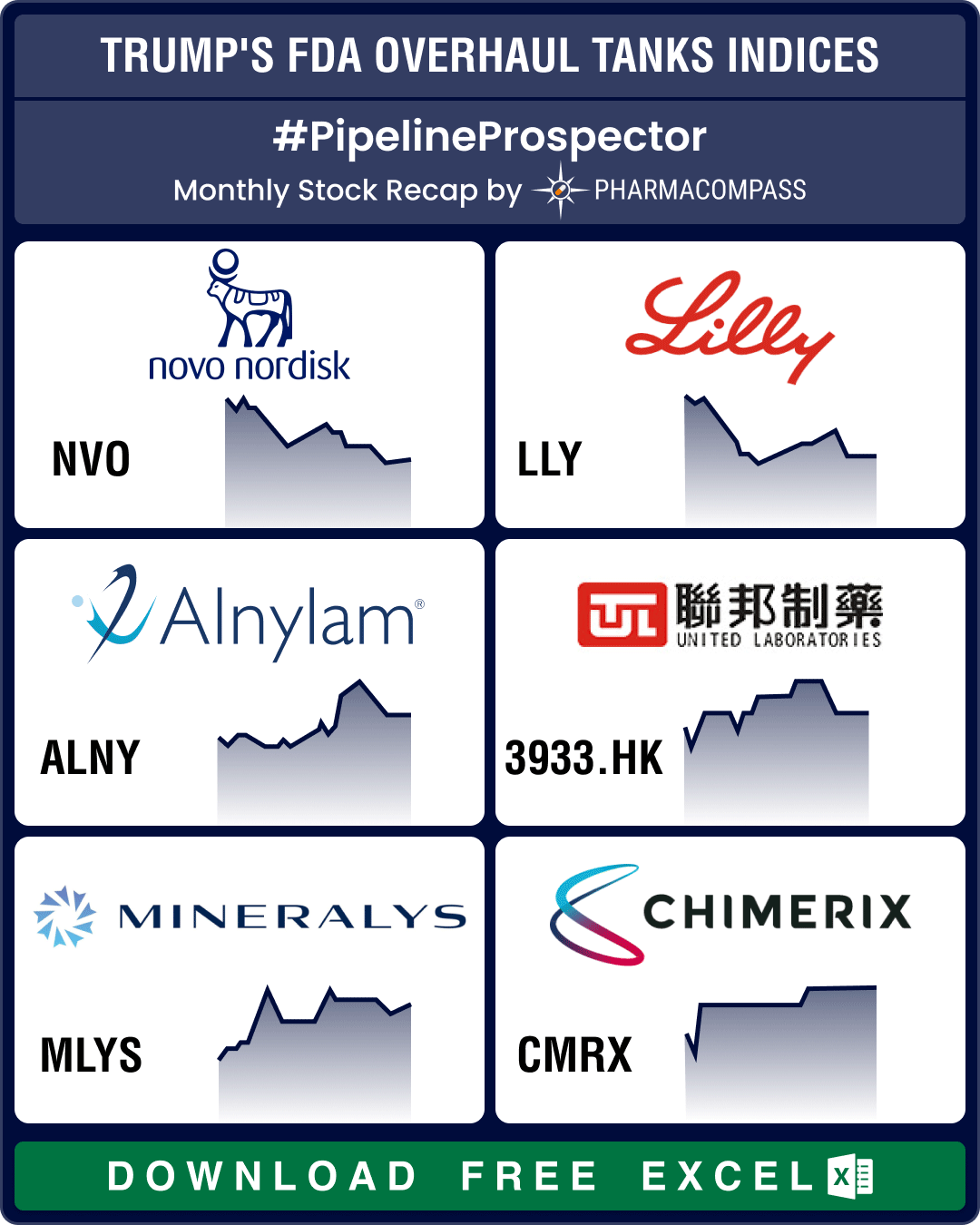
March ended with news that the top vaccine regulator of the US Food and Drug Administration (FDA), Peter Marks, was forced to resign. This impacted pharma indices adversely. The Nasdaq Biotechnology Index (NBI) dropped 6.22 percent from 4,525.73 in February to 4,244.37 in March. The SPDR S&P Biotech ETF (XBI) fell 8.61 percent from 88.74 to 81.10, and the S&P Biotechnology Select Industry Index (SPSIBI) declined 8.61 percent from 6,914.8 to 6,319.5.
The sector has been under pressure ever since US President Donald Trump announced plans to impose potential 25 percent tariffs on pharmaceutical imports. Just last week, Johnson & Johnson committed US$ 55 billion to set up four new manufacturing plants in the US. J&J’s announcement follows Eli Lilly’s US$ 27 billion US investment pledge announced in February. Merck too is deepening its domestic footprint with the inauguration of a new US$ 1 billion, 225,000-square-foot vaccine manufacturing facility in Durham, North Carolina, as part of its broader US $20 billion US investment strategy spanning 2018 to 2028.
Trump’s trade policy is also driving corporate decisions. In an opportunistic maneuver, Mallinckrodt and Endo — both emerging from bankruptcy due to opioid litigation — decided to merge via a US$ 6.7 billion deal that strategically leverages their extensive US manufacturing capabilities. The new company will focus on branded drugs, and hopes to benefit from tariffs on imported goods.
Access the Pipeline Prospector Dashboard for March 2025 Newsmakers (Free Excel)
Roche, AbbVie ink US$ 5.3 bn, US$ 2.2 bn deals for amylin-based obesity treatments; AstraZeneca strikes multiple deals
March saw competition in the obesity treatment market intensify with multiple pharmaceutical giants making strategic moves. The month witnessed a rise in interest in amylin-based treatments. Amylin is a peptide hormone that plays a role in regulating blood sugar and appetite. The amylin pathway has the potential advantage of reducing fat rather than lean muscle.
Roche signed a US$ 5.3 billion agreement with Zealand Pharma for petrelintide, a promising long-acting amylin analog administered weekly through a subcutaneous injection. Zealand will receive an upfront payment of US$ 1.65 billion from Roche.
AbbVie marked its entry into the lucrative obesity market through an up to US$ 2.23 billion licensing agreement with Danish biotech firm Gubra for GUB014295, another amylin-targeting drug.
Towards the end of the month, Novo Nordisk announced a US$ 1 billion licensing deal with Lexicon Pharmaceuticals for the latter’s experimental obesity drug. Under the terms of the deal, Novo will get worldwide rights to develop, manufacture and commercialize the drug, LX9851, for obesity and associated metabolic disorders.
Novo also secured ex-China rights to United Laboratories International Holdings’ UBT251 in a deal worth up to US$ 2 billion. This is a "triple-G" drug that targets three receptors — GLP-1, GIP, and glucagon — involved in regulating blood sugar and appetite.
Similarly, Merck entered into a US$ 2 billion-worth agreement with Jiangsu Hengrui Pharmaceuticals for HRS-5346, an oral drug targeting lipoprotein(a) reduction to prevent cardiovascular diseases.
In immunology, Sanofi’s US$ 1.9 billion acquisition of Dren Bio’s immunology unit centered around DR-0201, a novel treatment for autoimmune diseases that targets specific immune cells (B-cells) that mistakenly attack healthy cells. Early clinical studies suggest this approach could potentially reset the adaptive immune system, offering sustained remission for patients with refractory conditions like lupus.
AstraZeneca has been particularly active this month. Astra announced a US$ 1 billion acquisition of Belgian cell therapy biotech EsoBiotec and a US$ 1.35 billion agreement with Alteogen to develop subcutaneous formulations of its oncology drugs. The acquisition brings AstraZeneca the innovative ENaBL platform, which could transform cell therapy by reducing treatment times from weeks to minutes while significantly lowering manufacturing costs.
The British-Swedish drugmaker also announced collaborations with Chinese biotech firms, Harbour BioMed and Syneron Bio, as well as a joint venture with BioKangtai to develop, produce, and market vaccines for respiratory and other infectious diseases.
Oncology-focused biopharma Chimerix got acquired by Jazz Pharmaceuticals for US$ 935 million. Its lead candidate dordaviprone is currently under FDA review for treating H3 K27M-mutant diffuse glioma, a rare brain tumor.
Access the Pipeline Prospector Dashboard for March 2025 Newsmakers (Free Excel)
GSK’s Blujepa, Roche’s TNKase mark first breakthroughs in 30 years for uUTI, acute ischemic stroke treatments
The month saw two key FDA approvals — GSK’s Blujepa and Roche’s TNKase. GSK’s Blujepa (gepotidacin) is the first new class of oral antibiotics for uncomplicated urinary tract infections (uUTIs) in nearly three decades. The drug, approved for the treatment of uUTIs in female adults and patients aged 12 years and older who weigh at least 40 kg, has a novel mechanism of action that offers hope against certain antibiotic-resistant strains.
Similarly, the US approval of Roche’s TNKase (tenecteplase) for acute ischemic stroke marks the first new treatment option for stroke patients in nearly 30 years. Its five-second administration offers a critical advantage in time-sensitive stroke management.
Sanofi and Alnylam’s RNAi therapy Qfitlia (fitusiran) became the first approved treatment for both hemophilia A and B. And Novartis’ prostate cancer drug Pluvicto won an expanded approval to treat PSMA-positive metastatic castration-resistant prostate cancer (mCRPC) before taxane-based chemotherapy.
Alnylam Pharmaceuticals (stock up 9 percent in March) secured FDA approval for Amvuttra (vutrisiran) to treat cardiomyopathy of wild-type or hereditary ATTR-CM, making it the first medication approved in the US to address both ATTR-CM and polyneuropathy of hereditary transthyretin-mediated amyloidosis.
Access the Pipeline Prospector Dashboard for March 2025 Newsmakers (Free Excel)
Novo’s CagriSema underwhelms in late-stage trial; J&J’s med fails, serving another blow to depression treatment
March also saw several high-profile clinical disappointments. Novo Nordisk’s (stock down 23 percent) CagriSema (cagrilintide/semaglutide) delivered underwhelming results in its second late-stage obesity trial. While the drug demonstrated superior weight loss compared to Wegovy, showing 13.7 percent average weight reduction over 68 weeks, market expectations for a breakthrough superior to rival Eli Lilly’s Mounjaro (tirzepatide) were not met.The neurology segment continued to see disappointments, especially in the treatment of depression. J&J’s aticaprant failed in late-stage depression studies for major depressive disorder (MDD), forcing the company to abandon its development for this indication. The disappointing results come on the heels of similar setbacks within the same drug class, most notably with Neumora Therapeutics’ navacaprant.
Access the Pipeline Prospector Dashboard for March 2025 Newsmakers (Free Excel)
Our view
The pharmaceutical landscape has been undergoing dramatic changes since Trump moved into the White House for a second term. The most significant change for the sector is FDA’s overhaul. For decades, FDA has been the gold standard for drug regulation. The overhaul has changed that.“The FDA as we’ve known it is finished,” former FDA Commissioner Robert Califf said in a social media post. Healthcare investment bankers are already talking about large deals getting stalled as executives grapple with the volatile policy scenario. Given these uncertainties, our sentiments are bearish, at least for the short term.
Access the Pipeline Prospector Dashboard for March 2025 Newsmakers (Free Excel)
Pharma & Biotech Newsmakers in March 2025
| Company | Country | Currency | Market Cap (Bn) | Change In Market Cap (M) | Stock Price | Change In Price |
|---|
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
Image Credit : TRUMP'S FDA OVERHAUL TANKS INDICES by PharmaCompass license under CC BY 2.0
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”








