Artificial intelligence (AI) is changing the world and bringing
efficiencies across all industries. Pharmaceuticals is one of them. AI can
accelerate drug discovery, streamline clinical trials, and personalize
medicine. It holds the potential to revolutionize the pharma industry.It takes anywhere between 10 to 15 years and around US$ 3 billion to take a new drug from its
discovery phase to the market. AI can drastically cut both the timespan and
costs and bring life-saving yet affordable treatments to the market at a faster
pace. The global AI market in healthcare was estimated to be about US$ 21
billion in 2024 and is expected to grow to over US$ 148 billion by 2029, compounding at an annual
growth rate of 48.1 percent.Drugmakers are eager to ride the AI wave. Players like Bayer, Merck KGaA, Moderna, BMS, Roche, Astellas, Amgen, Eli Lilly, Sanofi, Novo Nordisk and Johnson & Johnson have announced
collaborations, signed deals and entered into partnerships in the AI space.
Many others, such as Exscientia, Insilico, Berg, Nimbus, Recursion, and Pharos iBio, are a step ahead and are holding clinical trials on drugs developed using AI.On its part, the US Food and Drug Administration (FDA) has
recognized the increased use of AI and machine learning (ML) and has reiterated its commitment to ensure the safety and efficacy of drugs “while facilitating innovations in their development.” To this end, the US agency
published a paper in March that lists out its
approach towards the use of AI.Novo, Lilly turn to AI for
drug discovery; AI-based firms sign multiple dealsWith increased adoption of wearable devices, e-health services and
other technology-driven offerings in medicine and healthcare, there is valuable data out there that drugmakers can analyze to get more accurate predictions of a drug’s effects on the human body. Such an analysis speeds up drug development and reduces side effects of therapies. Ergo, several large drugmakers have
signed crucial AI deals. For example, Novo Nordisk has inked a potential US$ 2.7 billion deal with Valo Health to discover and develop novel treatments for cardio-metabolic diseases. The collaboration between the two organizations will leverage the capabilities of Valo’s Opal Computational Platform, including access to real-world patient data, AI-enabled small molecule discovery and Biowire human tissue modeling platform designed to speed up the discovery and development process.Similarly, Eli Lilly has entered into a US$ 494 million deal with Fauna Bio to use the latter’s AI platform — Convergence — for preclinical drug discovery efforts in obesity. Convergence analyzes genomic data from 452 mammal species and various tissue types to
create a comprehensive dataset. By integrating human data and information from
animals with natural disease resistance, the platform can identify potential
drug targets.AI-based pharma firms like Isomorphic Labs and Biolojic are also landing multiple deals. Isomorphic is owned by Google’s parent Alphabet. Along with Google DeepMind, it has created a new AI model that can
accurately predict 76 percent of protein interactions with
small molecules. In January, Isomorphic inked deals with Novartis and Lilly for a combined
value of nearly US$ 3 billion.Biolojic Design uses computational biology and AI to transform
antibodies into programmable, intelligent medicines. Earlier this month, it
announced a multi-target drug discovery collaboration
with Merck KGaA, which includes
antibody-drug conjugates. Biolojic has also inked deals with Teva and Nektar recently.Amgen harnesses generative
biology for protein-based drugs; Sanofi ties up with OpenAIFinding a good protein drug candidate is like finding a needle in
a haystack. Drug developers typically start by looking at proteins in nature and then go through the painstaking process of shaping them into safe and effective drugs.Generative biology is a revolutionary approach to drug discovery
and development that leverages ML and AI to design novel protein therapeutics. Amgen is using generative biology to innovate new protein-based drugs that have desired structures and properties based on existing protein data inputs. In fact, the California-based multinational has said it is “integrating AI across all operational levels.” It has collaborated with PostEra to advance up to five small molecule programs.Amgen is building AI models trained to analyze one of the world’s largest human datasets on an NVIDIA full-stack data center platform, known as DGX SuperPOD, that will be installed at Amgen’s deCODE genetics’ headquarters in Reykjavik, Iceland. This system will be used to build a human diversity atlas for drug target and disease-specific biomarker discovery, providing vital diagnostics for monitoring disease progression and regression.AI can analyze individual data like genetic makeup,
lifestyle and medical history to come up with personalized therapies. Amgen is working on
AI-driven precision medicines, and potentially individualized therapies, at its
Iceland facility.Sanofi too has signed multiple deals in recent months. In May,
the French drugmaker signed a collaboration with ChatGPT maker OpenAI and Formation Bio to build AI-powered software to accelerate drug development. Sanofi is also collaborating with BioMap in a deal that could be
worth up to US$ 1 billion. The deal is expected to
enable superior prediction from limited data in a range
of therapeutic areas, including immunology, neurology, oncology, and rare
diseases.AI tools can create control
arms, digital twins and slash failure rate in clinical trialsTraditional ways of drug development are fraught with challenges,
and 90 percent of drug candidates in clinical trials tend to fail. Major reasons behind this are poor patient cohort selection and recruiting mechanisms, and the inability to monitor patients effectively during trials. AI tools like Trial Pathfinder study real data obtained from patients’ electronic health records and simulate clinical trials for the drug with different eligibility criteria. Trial Pathfinder also calculates trial hazard ratios, a scientific term that compares the survival rates of those given and not given the drug.AI and synthetic data can also be used to create control arms in
clinical trials, which can help speed up the process, reduce costs, and improve
the quality of data. For example, FDA has supported the use of a Medidata Synthetic Control
Arm in a
phase 3 trial of Medicenna’s MDNA55 in recurrent
glioblastoma.In addition to observing real-time patient data, researchers can
also create digital twins, virtual replicas of cells,
organs, or people, which they can use to simulate and predict various clinical
outcomes during a trial.Our viewThe
pharmaceutical industry is on the cusp of a revolution. In the coming years, we
hope to see some tangible results of the efforts being put in by drugmakers, AI
developers and regulatory agencies. The CPhI Annual Report 2023 has predicted
that in 2030, over half of FDA-approved drugs will involve AI in their development and/or manufacturing. The winner, in our view, will be the end-user who will have cheaper, safer and more effective treatments, delivered at a faster pace.
Impressions: 1659
Generic drugs play a crucial role in providing access to life-saving drugs at affordable prices. To that end, drugmakers submit Drug Master Files (DMFs) or their ‘recipes for making generics’ to the US Food and Drug Administration (FDA) for review. Of these, Type II DMFs involve active pharmaceutical ingredients (APIs) for both branded and generic drugs.PharmaCompass has been reviewing Type II
submissions for several years now. We have noticed that these filings have been
increasing in recent years. There has been a 33.5 percent
increase in Type II DMF submissions since Q1 2018 (when 176 Type II DMFs had
been submitted). However, at 235, the first quarter (Q1) of 2024 saw only a 1.3
percent increase in DMF submissions over Q1 2023. In Q1 2023, DMF submissions
had increased by 21.5 percent (over Q1 2022).Of the 235 Type II submissions received by the FDA in Q1 of this
year, only 35 (or around 15 percent) had their review completed under the Generic Drug User Fee Act (GDUFA). The total number of reviews completed by the US federal agency so far this year is 129.In all, FDA received 353 Type II, III, IV, and V DMF submissions,
compared to 291 in Q1 2023, an increase of 21.3 percent.View FDA DMF Filings in Q1 2024 (Power BI Dashboard, Free Excel Available)DMF submissions from India
dip 1.8%; China witnesses 42% rise in filings As has been the trend, India (dubbed as the pharmacy of the world) and China filed the maximum DMF submissions, with 107 and 101 submissions respectively. However, DMFs filed by Indian companies saw a marginal dip of 1.8 percent in Q1 2024 — in Q1 2023, Indian companies had filed 109 Type II DMFs. Meanwhile, DMF filings by Chinese drugmakers increased 42.2 percent in Q1 2024, up from 71 in Q1 2023.India’s MSN Laboratories was the clear leader with 15 DMF submissions to the FDA, while China’s Sichuan Elixir
Pharmaceutical came a distant second with nine submissions. Indian drugmakers Aurobindo Pharma and Global Calcium (with seven submissions each) were tied at third position. There were two players at the fifth spot — India’s Dr. Reddy’s Laboratories and China’s Zhejiang Jingsheng
Pharmaceutical — as both these drugmakers registered six submissions each.View FDA DMF Filings in Q1 2024 (Power BI Dashboard, Free Excel Available) Diabetes, cancer drugs emerge
as hot molecules in Q1 2024Approved in June 2020, triheptanoin is the first and only FDA-approved treatment for
children and adults with long-chain fatty acid oxidation disorders (LC-FAOD).
At five, triheptanoin saw the maximum DMF filings.
Four of those applications have already been positively reviewed.Anti-diabetic drug sitagliptin phosphate
monohydrate saw four DMF filings. Empagliflozin, the API found in Boehringer Ingelheim and Eli Lilly’s diabetes drug Jardiance, drew three DMF filings in Q1 2024.In the US, Jazz Pharmaceuticals’ Zepzelca (lurbinectedin) is indicated for the treatment of adult patients with metastatic small cell lung cancer (SCLC). Three DMF applications were submitted for lurbinectedin. Similarly, ruxolitinib phosphate, used for the treatment of
patients with intermediate or high-risk myelofibrosis, also received three DMF
applications.Semaglutide — the glucagon-like peptide-1 receptor agonist that catapulted Novo Nordisk to the position of the most valuable public company in Europe — received three DMF filings. Semaglutide’s arch rival, tirzepatide, also drew three DMF filings.AstraZeneca’s Brilinta (ticagrelor) plus aspirin is currently approved in
over 115 countries for the prevention of stroke, heart attack, and other events
in adults with acute coronary syndrome (ACS). Ticagrelor received three DMF
submissions. Vonoprazan fumarate, a first-in-class potassium-competitive acid
blocker to treat acid-related diseases, also received three DMF submissions.View FDA DMF Filings in Q1 2024 (Power BI Dashboard, Free Excel Available) Eighteen DMFs filed for first
time in Q1 2024 During the first quarter of this year, 14 drugs saw first time filing of DMFs. Together, these 14 drugs attracted 18 DMF filings with tirzepatide topping the list with three submissions from Chinese companies.The 14 drugs that saw first time filing were clascoterone, capmatinib hydrochloride, niraparib tosylate
monohydrate, vibegron, cabozantinib sulfate,
ruxolitinib hemifumarate, ripretinib, ruxolitinib, ruxolitinib mesylate, tucatinib hemiethanolate, tecovirimat, tirzepatide, tolvaptan povidone, and azilsartan.View FDA DMF Filings in Q1 2024 (Power BI Dashboard, Free Excel Available) Our viewUntil 2020, DMF submissions by Indian companies used to be double those of Chinese and American
firms put together. In 2022, DMF filings from India began to dip, while
submissions by Chinese companies began to rise. In the first half of 2023,
there was a 46.5 percent rise in DMF submissions from China. The gap between
DMF filings by India and China has narrowed down considerably this year. The
reasons behind this phenomenon may be multiple. But it definitely implies that
America will find it increasingly difficult to reduce its reliance on China.
Impressions: 2166
After a year when
drug approvals by the US Food and Drug Administration
(FDA) slipped to the lowest since 2016, the first half of 2023 (H1 2023) saw a
62.5 percent growth in drug approvals by the Center for Drug Evaluation and Research (CDER) as compared with the same period last year. FDA’s CDER approved 26 new drugs
in 2023, up from 16 in H1 2022. Approvals of biologics by the FDA surpassed full-year totals for many of the past 20 years. Nine new biologics were approved by FDA’s Center for Biologics Evaluation and Research (CBER) in H1 2023, as against just eight
for 2022.While FDA approved more drugs, authorizations by the European Medicines Agency (EMA) also increased but drug approvals by Health Canada dipped by approximately 50 percent. In the first half of 2023, EMA authorized 14 drugs, as opposed to 10 in H1 2022. Similarly, Health Canada approved 13 drugs against 25 in H1 2022.Among the
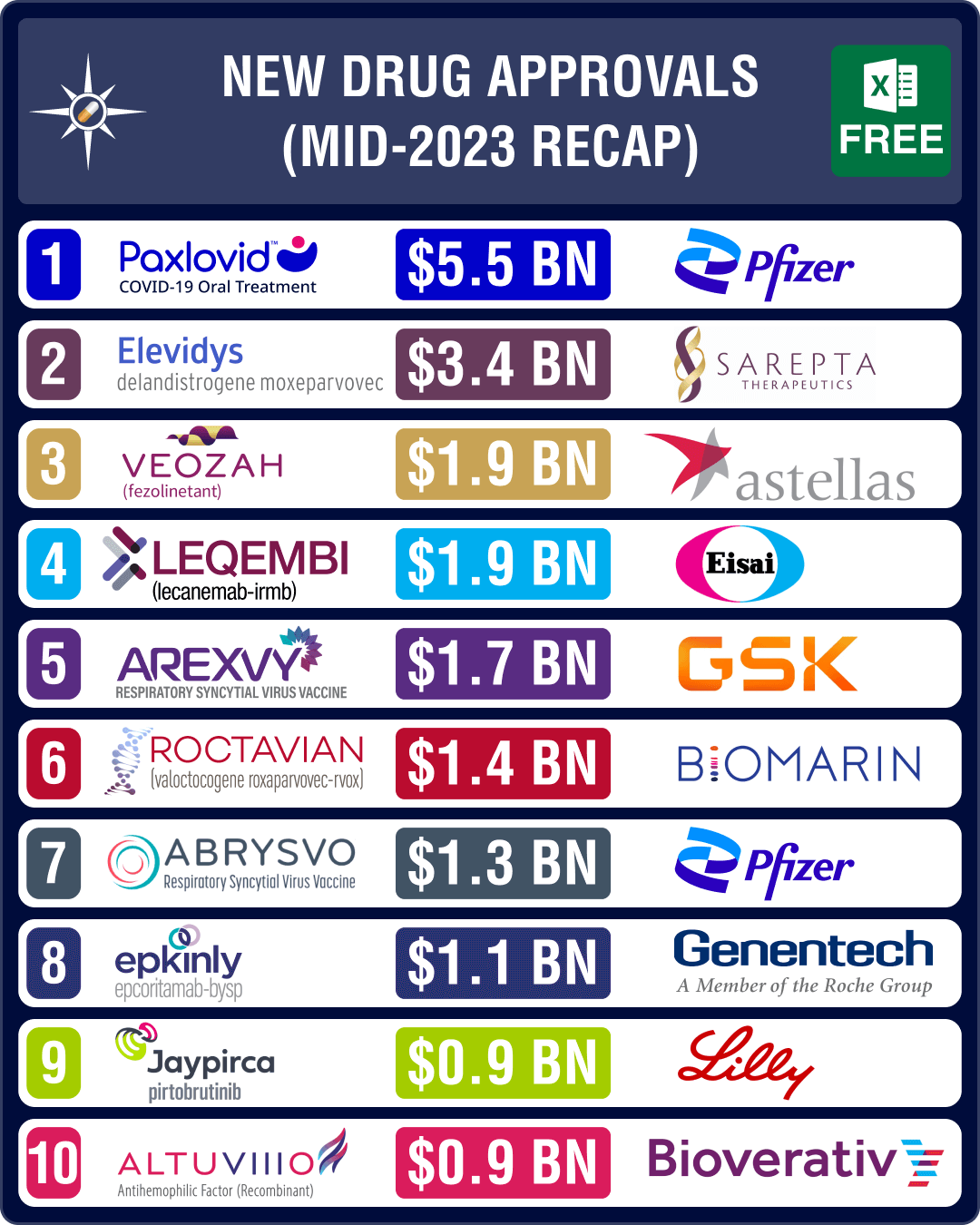
promising drugs approved in H1 2023 are Biogen and Eisai’s Alzheimer’s drug Leqembi (lecanemab), Astellas Pharma’s drug for hot flashes (associated with
menopause) Veozah, AbbVie and Genmab’s blood cancer drug Epkinly and, GSK and Pfizer’s RSV vaccines Arexvy and Abrysvo, respectively.View New Drug Approvals in first half of 2023 with Estimated Sales (Free Excel Available)GSK, Pfizer’s RSV vaccines to generate over US$ 1 billion by 2028The highlight of H1 2023 has been the approval of vaccines for respiratory syncytial virus (RSV), a contagious virus that causes infections of the respiratory tract, and can cause severe illness in older adults and infants. In May 2023, FDA approved GSK’s Arexvy, respiratory syncytial virus (RSV) vaccine for people aged 60 and older, making it the first vaccine approved in the US that protects against the disease. The drug is expected to generate US$ 1.73 billion in peak sales by 2028.Weeks later, the agency approved Pfizer’s Abrysvo, another RSV vaccine for older adults. The vaccine is expected to be available
in the third quarter of 2023. In late August, Pfizer also bagged an FDA nod for
use of Abrysvo to prevent RSV in infants by vaccinating pregnant women. The
drug is expected to generate US$ 1.31 billion in peak sales by 2028.View New Drug Approvals in first half of 2023 with Estimated Sales (Free Excel Available) Leqembi, Veozah, Epkinly likely to join ‘over US$ 1 billion club’ by 2028In March, the US health regulator suggested stricter trials for accelerated approval of cancer drugs. The proposed recommendation followed criticism over the approval of Biogen and Eisai’s Alzheimer’s treatment Aduhelm through this pathway in June 2021.Contrary to this
stand though, FDA had approved another Alzheimer’s drug from Biogen and Eisai — Leqembi (lecanemab) — through the same pathway in January 2023. Leqembi is an amyloid beta-directed antibody indicated for the treatment of Alzheimer’s disease. The drug is expected to generate US$ 1.9 billion in peak sales by 2028. Later in July, FDA expanded the accelerated approval of lecanemab to a traditional approval for early-stage Alzheimer’s disease.The US regulator
approved several potential blockbuster drugs during the month of May. It
granted full approval to Pfizer’s Covid-19 oral antiviral pill Paxlovid (nirmatrelvir and ritonavir) during the month, which had been
granted an emergency use authorization during the heydays of the pandemic. Paxlovid is the first oral antiviral pill for Covid-19. With the pandemic behind us, Paxlovid’s sales have been waning, though it is still projected to yield approximately US$ 5.5 billion in sales by 2028.The other
important drug approved in May is Japanese drugmaker Astellas Pharma’s oral drug Veozah (fezolinetant) for the treatment of moderate to severe vasomotor symptoms (VMS), or hot flashes associated with menopause. The drug will cost US$ 550 for a month’s supply. Veozah is expected to generate US$ 1.9 billion in sales by 2028.FDA also granted
accelerated approval to AbbVie and Genmab’s blood cancer therapy Epkinly (Epcoritamab-Bysp) in May. It
has bagged approval to treat patients with diffuse large B-cell lymphoma
(DLBCL), and is likely to emerge as a US$ 1.1 billion product.View New Drug Approvals in first half of 2023 with Estimated Sales (Free Excel Available) Sarepta, Biomarin’s gene therapies bag FDA nod; Lilly’s Jaypirca okayed for lymphomaIn June, FDA
approved Sarepta’s Elevidys and Biomarin’s Roctavian. Elevidys bagged accelerated approval in Duchenne muscular dystrophy (DMD)
patients aged four to five years. This is the first gene therapy approved by
the FDA for the treatment of certain patients with DMD. Elevidys is expected to
generate US$ 3.36 billion in peak sales by 2028.BioMarin’s one-time gene therapy – Roctavian – has been approved by the FDA to treat severe hemophilia A, a
rare bleeding disorder. Priced at US$ 2.9 million, Roctavian is the first gene
replacement therapy to be approved for the condition. Roctavian is expected to
generate US$ 1.43 billion in peak sales by 2028. This approval follows another
approval granted in November 2022 for the first gene therapy to treat adults with hemophilia B — CSL Behring and uniQure’s Hemgenix.In January, Lilly’s Jaypirca (pirtobrutinib) bagged FDA nod, thereby becoming the first and only non-covalent, BTK Inhibitor for the
treatment of adult patients with relapsed mantle cell lymphoma which did not
respond to other BTK Inhibitors. Jaypirca is expected to generate US$ 0.91
billion in peak sales by 2028.In March, FDA approved Pfizer’s Zavzpret (zavegepant) as the first and only calcitonin gene-related peptide (CGRP) receptor antagonist nasal spray for the acute treatment of migraine. Zavzpret is expected to generate US$ 0.649 billion in peak sales by 2028.Another important FDA approval was granted to GSK’s drug Jesduvroq — it is now the first oral treatment for anemia caused by chronic kidney disease in adults who have been on dialysis for at least four months.View New Drug Approvals in first half of 2023 with Estimated Sales (Free Excel Available) FDA rejects Lilly’s donanemab, Alvotech/Teva’s Humira biosimilarIn January, when
the FDA had approved Leqembi, the agency had rejected Eli Lilly’s bid for an accelerated approval pathway for its experimental Alzheimer’s disease drug donanemab. The agency had cited a lack of participants who received continuous treatment with donanemab for at least 12 months as the reason for its decision. However, Lilly
announced positive late-stage trial
results on donanemab in July — it slowed cognitive decline by 35 percent compared to a placebo in a late-stage trial. An FDA decision on the drug is likely around 2023-end or in early 2024.Similarly, FDA turned down Lilly’s mirikizumab for the treatment of ulcerative colitis. The agency also rejected a BLA for Eli Lilly’s ulcerative colitis drug mirikizumab over manufacturing concerns. Besides these, FDA also rejected Alvotech/Teva’s Humira biosimilar — AVT02 — for a second time this year.View New Drug Approvals in first half of 2023 with Estimated Sales (Free Excel Available) Our viewAfter the
pandemic, business is back to normal at the FDA. The number of CDER approvals
in the first half of this year are approximately 70 percent of the approvals in
2022. We anticipate more approvals in the upcoming months. If the FDA sustains
this pace, approvals this year could potentially double those of the previous
years.
Impressions: 3539
In 2022, the pandemic continued to bring in a windfall for
pharmaceutical companies manufacturing Covid-19 products, with Pfizer’s total annual sales reaching
a record high by surpassing US$ 100 billion for the first time.AbbVie, despite not having any Covid products
in its portfolio, maintained its second position due to the sales of its blockbuster drug Humira and other medicines. Johnson & Johnson held
onto the third spot, while Merck experienced the most significant
change, climbing to the fourth position from seventh place in the previous
year. AstraZeneca also moved up a spot to the
eighth position. On the other hand, Novartis (fifth), Roche (sixth), BMS (seventh) and Sanofi (ninth) slipped in the rankings,
while GSK (tenth) retained its position.
There were no new entries in the top 10 in terms of pharmaceutical sales.View Our Interactive Dashboard on Top Drugs by Sales in 2022 (Free Excel Available)Pfizer retains top position
as Comirnaty, Paxlovid, together generate over US$ 56 billion In 2022, Pfizer’s Covid-19 products — vaccine Comirnaty and antiviral pill Paxlovid — generated combined sales of US$ 56.7 billion. With a haul of US$ 37.8 billion, Comirnaty comfortably
maintained its position as the top-selling drug for a second year in a row.
Paxlovid brought in US$ 18.9 billion in its first full year on the market, grabbing the number four spot.Moderna’s messenger RNA
Covid vaccine Spikevax generated US$ 18.4 billion in revenues, emerging as the
fifth largest drug by sales in 2022.View Our Interactive Dashboard on Top Drugs by Sales in 2022 (Free Excel Available) AbbVie’s Humira, J&J’s Stelara post impressive growth; BMS face revenue declineAbbVie’s cash cow Humira, the bestselling non-Covid
product in biopharma history, achieved sales of US$ 21.24 billion (partner Eisai reported another US$ 370 million) in 2022. While Humira brought in approximately 37 percent of the company’s total net revenues of US$ 58 billion in 2022, strong revenues from blood cancer drug Imbruvica, psoriasis treatment Skyrizi and rheumatoid arthritis med Rinvoq also contributed to AbbVie’s strong performance.J&J generated US$ 52.56 billion in 2022
revenues. Its pharmaceutical segment experienced an operational growth of 6.7 percent, driven by impressive
performance of key drugs such as Stelara for inflammatory diseases
and Darzalex for multiple myeloma.
Stelara, which generated US$ 10.2 billion in overall sales last year (a
growth of 6.9 percent), is expected to face
generic competition in the US as its
exclusivity is ending in 2023.BMS’ sales remained relatively
unchanged even though its blockbuster multiple myeloma drug, Revlimid, experienced a 22 percent
revenue decline due to generic competition. While Revlimid still generated
sales of US$ 9.98 billion (partner BeiGene reported another US$ 80 million) in 2022, two other drugs, anticoagulant Eliquis and cancer drug Opdivo performed well. Eliquis
generated US$ 11.8 billion in sales (partner Pfizer reported another US$ 6.5
billion), a 9.2 percent increase over 2021, while Opdivo achieved US$ 8.25 billion in sales (partner Ono Pharmaceuticals reported another US$ 1.05 billion). The drugmaker also introduced new products like Opdualag, Abecma and Reblozyl, which bolstered
its overall sales.View Our Interactive Dashboard on Top Drugs by Sales in 2022 (Free Excel Available)Merck expands use of blockbuster Keytruda; oncology drugs boost Astra’s revenueMerck’s blockbuster cancer drug Keytruda had a remarkable year,
achieving a significant year-on-year growth of 22 percent and generating US$ 20.9 billion in 2022 sales. This success propelled Keytruda to the third position among the top-selling drugs. Keytruda’s continuous expansion into new indications and treatment lines has solidified its position as a leading PD1 inhibitor, and is expected to maintain its momentum in 2023. Additionally, Merck’s Covid-19 pill Lagevrio recorded impressive sales of US$ 5.7 billion in 2022. Overall, at US$ 52 billion, the drugmaker posted spectacular
growth in sales of 22 percent.AstraZeneca also experienced an
impressive growth of 18 percent in 2022 to reach US$ 43 billion in revenue. The
growth was primarily driven by the success of its cancer treatments, with
blockbuster drugs like Tagrisso, Farxiga, Imfinzi, Lynparza playing a vital role. These drugs accounted for 35 percent of AstraZeneca’s overall revenue.View Our Interactive Dashboard on Top Drugs by Sales in 2022 (Free Excel Available)Newer
meds bolster Roche’s growth; Sanofi thrives with DupixentIn 2022, Roche experienced a sales growth of 2 percent at Constant Exchange Rate (CER) to reach CHF 45.55 billion (US$
49.8 billion). The company’s strong pharmaceutical sales were fueled by increasing demand for newer medicines, including Ocrevus (for multiple sclerosis), Hemlibra (for hemophilia A), Vabysmo
(for eye conditions), Evrysdi (for spinal muscular
atrophy) and cancer drug Tecentriq. In 2022, Sanofi recorded global sales of €43 billion (US$ 47.1 billion), representing a 7 percent growth at CER. The French drugmaker’s strong performance was driven by major drugs, particularly Dupixent and its vaccine franchise.
Dupixent brought in impressive sales of €8.3 billion (US$ 9.1 billion), marking a significant 57 percent increase over the previous year. Originally approved by the FDA in 2017 for atopic dermatitis,
Dupixent has expanded its applications to include moderate to severe asthma and
eosinophilic esophagitis, contributing to its continued growth. Sanofi expects
Dupixent to reach €10 billion (US$ 10.7 billion) in sales
in the current year.In July 2022, GlaxoSmithKline changed its name to GSK and demerged its consumer
healthcare business to form Haleon, thereby becoming a fully
focused biopharmaceutical company. The British drugmaker reported a modest 8 percent growth in revenue — at £29.32 billion (US$ 36.15 billion).View Our Interactive Dashboard on Top Drugs by Sales in 2022 (Free Excel Available)Our viewAs the pandemic wanes, 2023 could be a year of transition for many
drugmakers. Companies such as Pfizer, Moderna and AbbVie are implementing
significant changes to their business strategies. Pfizer foresees a substantial
decline in revenue, projecting a sharp drop of 33 percent with the reduced demand for
its Covid-19 products. AbbVie, on the other hand, anticipates a decline in
sales of its cash cow Humira due to increasing competition from biosimilars.
These drugmakers are turning to innovative therapies, new indications,
cost-cutting measures, acquisitions and partnerships to restrict the
anticipated drop in revenues and ensure sustained growth.Barring these drugmakers, several other big players shared
positive growth in the first quarter of 2023. While analysts predict Keytruda
and Dupixent to continue their strong growth, there is optimism surrounding diabetes and obesity drugs,
with Novo Nordisk’s Wegovy experiencing unprecedented
demand. Eli Lilly is also anticipating the US
approval of Mounjaro for obesity later this year.
All in all, we anticipate more changes in our list of top companies and drugs
for 2023!
Impressions: 5298
Every year, the list of top pharmaceutical products and companies by sales sees some churn. But the year 2021 was a lot different — it saw the pharma industry landscape change dramatically. It was a
year when the industry was busy developing vaccines and therapies so that the
world could recover from the Covid-19 pandemic. And this resulted in many
drugmakers raking in billions of dollars in sales.
As a result,
the top company of 2020 in terms
of pharmaceutical sales — Roche — slipped to the number five spot, while Pfizer, which was at number eight in 2020 after
spinning off its generic business, moved up to the number one slot.
The year
proved to be a good one for pharmaceutical companies.
Interestingly, last year none of the top 20 pharmaceutical companies saw a decline in
their revenue.
View Our Interactive Dashboard on Top Drugs by Sales in 2021 (Free Excel Available)
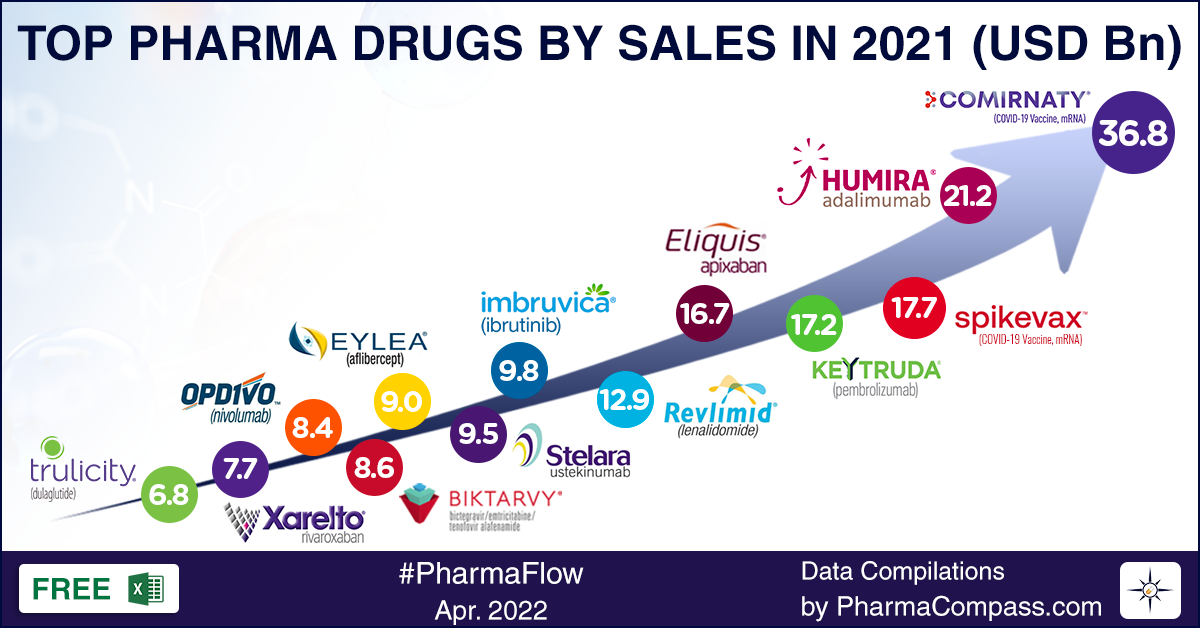
Pfizer’s Comirnaty steals the show
The
company that reaped the maximum gains from its Covid vaccine was Pfizer.
Comirnaty (tozinameran) was the top selling pharmaceutical
product of 2021, posting global revenues of US$ 36.8 billion. This
messenger-RNA Covid-19 vaccine, developed along with its German partner BioNTech, catapulted Pfizer to the slot of the top company by sales in 2021. Pfizer’s global topline grew from US$ 41.7 billion in 2020 to US$ 81.3 billion in 2021.
In
2020, Pfizer was at
number eight, behind Roche, Novartis, GSK, AbbVie, J&J, Merck and BMS. In 2021, it took a lead of billions of dollars on all these companies. The second largest drug company by sales — AbbVie — was way down at US$ 56.1 billion in global revenues. In fact, Comirnaty has
become the fastest-selling drug in the history of the pharmaceutical
industry.
Back in
December 2020, when both Comirnaty and Moderna’s Spikevax had bagged the US Food and Drug Administration’s emergency use authorization (EUA), there was a lot of uncertainty around how the promised doses would be delivered across the world. But both Comirnaty and Spikevax have proven to be a resounding success. Spikevax emerged as the third largest selling pharmaceutical product of 2021, bringing in US$ 17.7 billion for Moderna.
Analysts
expect both Pfizer-BioNTech and Moderna to sell even more vaccines in
2022. The reasons are manifold. First, SARS-CoV-2 is able to mutate often, and
is unlikely to be eradicated in the near future, creating a need for booster
shots. Second, the younger age groups are still to get vaccinated.
Along with
Comirnaty, Pfizer is battling Covid-19 with its antiviral pill, Paxlovid. Though the sales of Paxlovid have nosedived of late, Pfizer expects
Comirnaty and Paxlovid to help the New York-headquartered drug behemoth achieve US$ 100 billion
in 2022 revenues.
View Our Interactive Dashboard on Top Drugs by Sales in 2021 (Free Excel Available)
AbbVie
moves up, sans Covid product; Roche slips to number five
With no
Covid-19 related products, AbbVie did fairly well in 2021 — it moved up from the number four spot in 2020 to number two position, thanks to its Allergan acquisition, cash cow Humira (adalimumab), the continued success of its
cancer drug Imbruvica (ibrutinib) and an increase in sale of its psoriasis treatment Skyrizi (risankizumab) by a whopping 85 percent in 2021. Humira brought in sales of US$ 21.2 billion for AbbVie in 2021. However, things may change soon, with biosimilars of Humira slated to enter the market in 2023. The years 2022 and 2023 are likely to be transition years for AbbVie, as it works to build the market for its Humira successors — Rinvoq and Skyrizi.
Roche
emerged as a big loser in 2021, as several copycats of its blockbuster drugs
hit the market. Copycats to Roche’s three blockbuster cancer drugs—Avastin, Herceptin and Rituxan—eroded US$ 4.9 billion (CHF 4.73 billion) from the company’s sales in 2021. A large chunk
of growth for Roche came from its multiple sclerosis med Ocrevus, hemophilia drug Hemlibra, inflammatory disease therapy Actemra and PD-L1 inhibitor Tecentriq. The pandemic resulted in
lower-than-expected sales of Ocrevus (ocrelizumab) due to fears around its
immunosuppressive effects.
Like Roche, Novartis also slipped last year. It fell from
number two in 2020 to the number four slot in 2021. Essentially, Novartis is
struggling with a relatively lackluster pipeline. It had sold its 33 percent stake in Roche last
year for US$ 20.7 billion. It plans to use that sum for acquisitions in order
to beef up its pipeline. The Swiss drugmaker has also drawn up a restructuring
plan that includes layoffs of thousands of employees.
Before the
pandemic, Merck’s Keytruda was touted as the drug that would overtake
Humira at the top in 2024. The checkpoint inhibitor has continued to grow
impressively, adding new indications and treatment lines. Keytruda is now used
in close to 40 indications. With US$ 17.2 billion in sales, Keytruda emerged as
the fourth largest selling drug of 2022.
Overall
though, Merck slipped from number six to the number eight slot. This was due to
the fact that Merck had spun out its women’s
health, biosimilars and established brands businesses into Organon. However, its Covid-19 antiviral pill — Molnupiravir — was able to compensate for the lost revenue. Though the FDA is yet to grant the drug a full approval (it bagged an EUA in December 2021), advance sales agreements helped it rack up US$ 952 million in sales in the fourth quarter.
View Our Interactive Dashboard on Top Drugs by Sales in 2021 (Free Excel Available)
BMS moves
up with Eliquis, Revlimid; J&J lands at number three
Bristol Myers Squibb (BMS) moved up from number seven in 2020 to number six, thanks to two of its drugs that made it to top 10 — anticoagulant Eliquis at number five and oncology drug Revlimid at number six. However, Revlimid will soon face competition — four generic companies now have the approval to sell their versions of Revlimid (lenalidomide) after March 2022. Revlimid
sales are expected to drop from US$ 12.9 billion to just US$ 2.06 billion in
2026.
BMS posted
US$ 46.4 billion in global revenues, a nine percent increase from US$ 42.5
billion reported in 2020. In immuno-oncology, Opdivo
brought in US$ 7.52
billion in sales, while Yervoy drew in sales of US$ 2 billion
(an increase of 20 percent).
J&J’s pharma division brought in US$ 52.1 billion in revenues
last year, an increase of 14 percent over its revenues of US$ 45.6 billion
posted in 2020. Drugs like Darzalex (for multiple myeloma), Stelara and its Covid-19 vaccine brought in growth during 2021,
helping J&J move up from number five to the number three slot. J&J’s Covid-19 vaccine brought in US$ 2.4 billion in
sales.
View Our Interactive Dashboard on Top Drugs by Sales in 2021 (Free Excel Available)
GSK bags
approval for shingles vaccine; Takeda suffers setbacks
GlaxoSmithKline (GSK) slipped four places — from number three in 2020 to number seven in 2021. Though GSK did not have a drug in the top 10, sales of GSK and Vir Biotechnology’s Covid-19 antibody treatment
sotrovimab helped produce a seven percent increase in its 2021 revenue. The British drugmaker also bagged a critical FDA approval — its vaccine to prevent shingles (herpes zoster), Shingrix, bagged the agency’s nod in July. GSK hopes to double the sales of Shingrix by 2026.
GSK is also undergoing a major transformation, and plans to demerge its consumer health unit this year. The unit generated revenues of £9.6 billion (US$ 13 billion) last year, and GSK sees the demerger as a necessary step to fuel growth through the development of new vaccines and specialty medicines.
Sanofi managed to retain its ninth slot, even as
its global turnover increased from US$ 39.3 billion (Euro 36.04 billion) to US$
41.6 billion (Euro 37.76 billion). It snapped up Kymab, Tidal Therapeutics, Translate Bio, Kadmon Holdings, Origimm Biotechnology and Amunix in deals that bolstered its presence in immunology, immuno-oncology and vaccines. Dealmaking is on the French drugmaker’s menu for 2022 and beyond, Sanofi’s CFO said at this year’s virtual JP Morgan Healthcare Conference.
AstraZeneca’s global revenues grew from US$ 26.6 billion in 2020 to US$ 37.4 billion in 2021. However, its rank fell from nine in 2020 to 10 in 2021.
AstraZeneca
wrapped up the US$ 39 billion acquisition of Alexion in July 2021. Alexion’s rare disease franchise—led by C5 inhibitors Soliris and Ultomiris—added an extra US$ 3.1 billion to Astra’s top line last year.
Takeda suffered several clinical and regulatory setbacks in 2021, which it labeled as an “inflection year.” For Gilead, sales of its Covid-19 antiviral Veklury brought in US$ 5.6 billion last year,
helping its revenues grow by 11 percent.
View Our Interactive Dashboard on Top Drugs by Sales in 2021 (Free Excel Available)
Our view
If anything,
the pandemic has taught us that change is the only constant. It has also taught
us that products can become blockbusters in a matter of a few months.
The industry
landscape continues to change. On the one hand, we are seeing people scrambling
to get Covid vaccines and booster shots, on the other hand, the FDA has limited the use of monoclonal antibodies, such as Eli Lilly’s bamlanivimab and etesevimab and
Regeneron’s REGEN-COV (casirivimab and
imdevimab), in treating Covid caused by the Omicron variant. The FDA has also pulled the authorization
granted to GSK and Vir Biotechnology’s antibody therapy this month,
citing data that suggested it was unlikely to be effective against the dominant
Omicron sub-variant.
And last
week, there was news that demand for Pfizer’s
antiviral pill Paxlovid has remained unexpectedly low. The supply
of Paxlovid, which reduced hospitalizations or deaths in high-risk patients by
around 90 percent in a clinical trial, has far outstripped demand in many
countries like the US, the UK and South Korea.
Though Pfizer is hopeful of crossing US$ 100 billion in revenue this
year, much depends on how the pandemic pans out and what new research has to
say about the novel coronavirus. A lot will change once the pandemic becomes endemic.
The first four months of 2022 tell us that vaccines like Comirnaty and Spikevax
will continue to perform well.
But two years down the line, our charts could look very different.
Impressions: 8048
In 2016, M&A deals
fell drastically in both numbers and value. One key reason was the falling
through of the Pfizer-Allergan mega merger due to America’s crack down on inversion deals.
The year 2015 went down in
history as a record year for mergers and acquisitions (M&As) in the
pharmaceutical and biotech space, when deals worth US $ 300 billion were
announced.
The
highlight of 2015 was the mega-merger announced between US drugmaker Pfizer and
Ireland-based Allergan – the biggest-ever pharma transaction that
was worth more than US$ 160 billion.
2016 saw Pfizer and Allergan walk away from their US$ 160 billion merger when the new US
Treasury rules cracked down on inversion deals that were encouraging US
companies to move overseas to cut taxes.
The merger would have allowed
New York-based Pfizer to cut its tax bill by an estimated US$ 1 billion
annually by domiciling in Ireland, where tax rates are lower.
M&A deals dip by 20
percent in 2016
Although the Pfizer-Allergan
mega-merger did not go through, The Pharma Letter tracked transactions through the year and noted that although “worldwide merger and acquisition activity in the pharmaceutical and biotechnology sector in 2016 was plentiful”, the numbers and values were “well down” on those seen in the previous two years.
The number of transactions announced in 2016 was 130, compared to 166 M&A deals in 2015 – which was a record year – and 137 in 2014, says The Pharma Letter.
Values of the top 10 deals
drop to a third
The other crucial fact about
M&A deals in 2016 was that transactions that exceeded the US$ 1 billion
mark were down to just 23 in 2016, as against 30 in 2015 and 26 in 2014.
The Pharma Letter quotes a
KPMG report published earlier this year which notes that the total value of the
top 10 completed deals in the first half of 2016 amounted to US $ 67.2 billion
as opposed to US $190.4 billion in first-half 2015.
Sanofi-Actelion — the deal that wasn’t
Sanofi made headlines, not for the acquisitions it made, but for the ones it wasn’t able to close.
Late last year, the French
pharma giant was widely identified as the big player that managed to push Johnson & Johnson away from negotiations with Actelion only to lose its US$ 30 billion bid to J&J even though it would have delivered “approximately equivalent value to Actelion’s shareholders”.
The Actelion loss came after Sanofi was out bid by Pfizer for Medivation.
Pfizer agreed to buy the US
cancer drug company for US$ 14 billion in cash, adding its blockbuster prostate
cancer drug Xtandi to
the company's growing oncology roster.
Additionally, Pfizer acquired Anacor for US$ 5.2 billion to add an eczema gel to its portfolio.
Bayer’s US$ 66 billion takeover
The biggest deal announced in
2016 was Bayer’s US$ 66 billion takeover of the US seeds company Monsanto after months of wrangling. It was the German drug and crop-chemical company’s third offer that clinched the deal, which is also known to be the largest all-cash deal on record.
This signature
deal has disrupted the agribusiness sector, which in recent years has been
involved in a consolidation race largely triggered by factors such as shifting
weather patterns, intense competition in grain exports and a souring global
farm economy.
Top pharma companies by sales
Bayer’s acquisition of Monsanto makes the ranking of top pharmaceutical companies consistently complicated since Bayer will generate more sales from its crop
science and high-tech polymer division than from the sale of prescription
drugs.
Should divisions like
diagnostics, animal health, vaccines, consumer health be counted while
determining the size of a pharmaceutical company?
In a volatile global world,
where wild exchange rate fluctuations play their own major role in determining
the size of organizations, this week PharmaCompass
shares the revenues, as presented in the 2016 annual reports of top 15
companies, so that you can draw your own conclusions on the top drug companies
of the world.
Company Name
Currency
Pharma
Consumer Health
Medical Devices/ Diagnostics
Vaccines
Animal Health
Other Revenues
Total Sales
1
Pfizer
USD
41,600
3,407
6,071
1,746
52,824
2
Novartis
USD
48,518
48,518
3
Roche
CHF
41,047
11,589
52,636
4
Merck & Co.
USD
29,360
5,791
3,478
1,178
39,807
5
GlaxoSmithKline
GBP
16,104
7,193
4,592
27,889
6
Johnson & Johnson
USD
33,464
13,307
25,119
71,890
7
Sanofi
EUR
22,932
3,330
4,577
2,708
274
33,821
8
Gilead
USD
29,953
437
30,390
9
Abbvie
USD
25,560
78
25,638
10
Bayer
EUR
16,420
6,037
1,523
22,789
46,769
11
Amgen
USD
21,892
1,099
22,991
12
Astrazeneca
USD
21,319
1,683
23,002
13
Teva
USD
20,664
1,239
21,903
14
Eli Lilly
USD
18,064
3,158
21,222
15
Bristol-Myers Squibb
USD
17,702
1,725
19,427
Sales figures are reported in millions.
Currency exchange rate used CHF: 0.99 USD/ EUR: 1.06 USD / GBP: 1.25 USD
Ranking methodology
When it came to ranking
companies, based on their total sales, we at PharmaCompass did
not face any challenges while including the sales of prescription drugs along
with those of vaccines.
But matters got a little
complicated when we got down to ranking consumer health divisions.
For instance, while we have
included consumer health divisions of companies like Sanofi, GSK and
Bayer, which primarily sell OTC drug products (such as brands like Allegra, Voltaren and Aleve), we have excluded those of companies like Johnson
& Johnson, given their focus on baby and beauty products.
Such a demarcation — based on the focus of the company — will always be a matter of debate.
Similarly, revenue generated
from the sale of medical devices/diagnostics as well as revenues of animal health divisions were not included in our rankings.
In the case of companies like Bayer, whose Covestro’s division has over US$ 10 billion in sales from customer industries such as automotive, construction, electrical and electronics, and furniture, such sales were accounted for in ‘other revenues’.
Our table highlights the sales
revenue of various divisions of companies in order to bring more clarity into the
figures which were included in our rankings.
Impressions: 13636