The world of pharmaceuticals and biotechnology continued to evolve
this year with strategic alliances reshaping industry contours. With mid-size
deals taking centerstage, the growth trajectory appears to be marked by a
balance of both caution and calculated ambition.The deal-making environment was robust in 2023, with over 2,000
unique pharma and biotech deals totaling more than US$ 410 billion, according
to the PharmaCompass database. Last year, there were over 200 mergers and acquisitions (M&As) with transactions exceeding US$ 160 billion in total value. Oncology, infections and infectious diseases, and neurology had emerged as the top three therapeutic areas for deals.PharmaCompass’ analysis indicates that the momentum has been maintained in 2024. As of August 6, the industry had seen over 1,200 unique deals valued at more than US$ 230 billion, including over 120 M&A transactions surpassing US$ 60 billion in aggregate value. While oncology maintains its lead position, neurology and immunology have gained notable traction.The one big difference is that 2024 is yet to witness a mega-deal,
comparable to Pfizer’s US$ 43 billion acquisition of Seagen in 2023 or Amgen’s US$ 27.8 billion Horizon buyout announced in 2022. The largest transaction
thus far has been Novo Nordisk Foundation’s US$ 16.5 billion acquisition of Catalent, a contract development and manufacturing organization (CDMO).This
compilation does not include deals related to acquisition of facilities,
divestment, medical devices, diagnostics and animal health. We have considered
deals announced, irrespective of when these transactions were completed. For a comprehensive overview of CDMO deals and developments
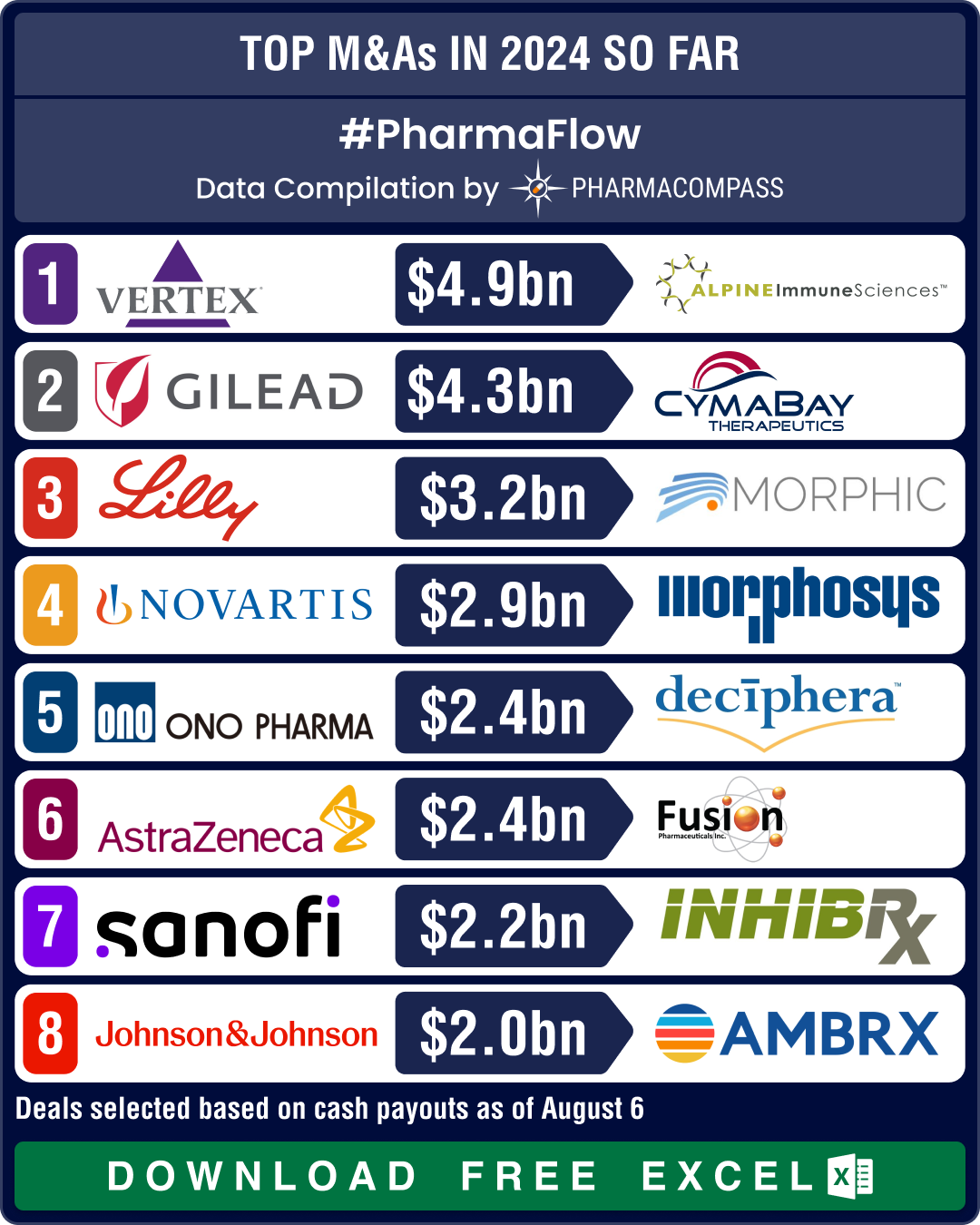
in 2024, please refer to our dedicated roundup.View Pharma & Biotech Acquisitions, Deals & Agreements in 2024 as of Aug. 6 (Free Excel Available) Vertex buys Alpine Immune for US$ 4.9 bn; Gilead’s CymaBay buyout pays off via FDA nodAmongst the biggest acquisitions of 2024 was Vertex Pharmaceuticals’ buyout of Alpine Immune Sciences for US$ 4.9 billion. It granted Vertex access to protein-based immunotherapies, including the promising povetacicept for IgA nephropathy, a serious kidney disease.Gilead Sciences’ acquisition of CymaBay Therapeutics for US$ 4.3 billion in February secured it
access to seladelpar (Livdelzi), an experimental drug that received FDA’s accelerated approval this month for primary biliary cholangitis, a liver disease that affects the bile ducts. Eli Lilly bolstered its presence in
the US$ 26.65 billion inflammatory bowel disease (IBD) market by purchasing Morphic Holding for approximately US$ 3.2 billion in July. Through this deal,
Lilly gained the oral IBD therapy candidate MORF-057, which will offer a more
convenient dosing option compared to injectable drugs currently available in
the market. Lilly sees the IBD space as a way to diversify beyond obesity.Merck expanded its ophthalmology portfolio by acquiring Eyebiotech Limited
for US$ 1.3 billion (plus US$
1.7 billion in milestone payments), obtaining Restoret for diabetic macular edema and neovascular age-related macular degeneration.Japanese drugmaker Ono Pharmaceutical acquired Deciphera Pharmaceuticals for US$ 2.4 billion, gaining Qinlock for
gastrointestinal stromal tumors and vimseltinib for tenosynovial giant cell
tumors. Sanofi targeted rare diseases by
purchasing Inhibrx for up to US$ 2.2 billion. The acquisition gave the French drugmaker access to INBRX-101 for Alpha-1 antitrypsin deficiency, a genetic condition that can cause lung and liver damage.View Pharma & Biotech Acquisitions, Deals & Agreements in 2024 as of Aug. 6 (Free Excel Available) Novartis buys two oncology firms for their assets; J&J, Genmab join ADC bandwagonNovartis has been on a shopping
spree, and has made two significant purchases this year. First, it acquired MorphoSys for € 2.7 billion (US$ 2.9 billion), thereby
adding the promising bone-marrow cancer treatment pelabresib to its pipeline. Second, it
announced the acquisition of Mariana Oncology for US$ 1 billion upfront (plus US$ 750 million in milestone payments), thereby expanding into radioligand
therapies (RLTs) to treat cancers with high unmet need. RLTs take a
targeted approach, delivering radiation to the tumor, while limiting damage to
the surrounding cells.AstraZeneca entered the field of
radioconjugates, which is a promising modality in the treatment of cancer, by
acquiring Fusion Pharmaceuticals for US$ 2.4 billion.In January this year, Johnson & Johnson had announced the acquisition of
antibody-drug-conjugate (ADC) developer Ambrx Biopharma for about US$ 2 billion. With this buyout, J&J has joined the likes of Bristol Myers Squibb, AbbVie and GSK who had entered this
promising field through acquisitions last year.Similarly, Denmark’s Genmab bought ProfoundBio for US$ 1.8 billion in cash, boosting its
oncology portfolio with three next-generation ADC candidates. This includes Rina-S, which recently received FDA’s fast track designation for the treatment of ovarian cancer.View Pharma & Biotech Acquisitions, Deals & Agreements in 2024 as of Aug. 6 (Free Excel Available) Novartis signs multiple
collaborations; GSK, Takeda, AbbVie sign billion-dollar dealsNovartis was not just busy signing M&A deals, it also signed a bevy of collaboration agreements. For instance, Shanghai-based Argo partnered Novartis on two
early-stage RNA interference candidates for cardiovascular diseases,
potentially earning the former up to US$ 4.2 billion plus tiered royalties.Novartis also agreed to pay up to US$ 3 billion (including US$ 150 million
upfront) to Dren Bio to use the latter’s Targeted Myeloid Engager and Phagocytosis platform to develop bispecific antibodies to treat cancer.Moreover, the Swiss drugmaker expanded its peptide discovery
collaboration with Japan-based PeptiDream in a deal worth over US$ 2.71 billion in milestone payments, plus an upfront payment of US$ 180 million. Peptide-drug conjugates (PDCs) are the next generation of targeted therapeutic drugs after ADCs and Novartis is, thus far, the only big pharma with FDA-approved radioligand PDCs. GSK entered a groundbreaking
partnership with Flagship Pioneering, potentially worth over US$ 7 billion, to identify and develop 10
novel drugs and vaccines. The deal, starting with respiratory and immunology
drugs, involves US$ 720 million in upfront and milestone payments for each candidate. This collaboration leverages Flagship’s extensive portfolio of over 40 biopharma companies with drug development capabilities.There were two significant deals in the field of neuroscience.
First, Takeda said it is paying Swiss
biotech AC Immune US$ 100 million upfront with potential further payments of US$ 2.1 billion for an exclusive option to license global rights to an Alzheimer’s vaccine and related immunotherapies.Second, AbbVie and clinical stage biotech Gilgamesh Pharmaceuticals joined forces in a deal
potentially worth over US$ 2 billion to develop a new class of psychedelic compounds for psychiatric conditions, combining AbbVie’s psychiatric expertise with Gilgamesh’s innovative neuroplastogen research platform.View Pharma & Biotech Acquisitions, Deals & Agreements in 2024 as of Aug. 6 (Free Excel Available) Our viewDuring this year, companies like Novartis (with US$ 16.8 billion), GSK (US$ 14.5 billion), Sanofi (US$ 11.9 billion), Bristol Myers Squibb (US$ 11.6 billion), and AbbVie (US$ 9.1 billion) have made
substantial investments in acquisitions, collaborations and other forms of
dealmaking.Though the deal-making environment is robust, we notice a shift towards mid-size transactions. Alongside, we notice a growing interest in areas such as ADCs, radiopharmaceuticals, and protein-based immunotherapies, underscoring their growing importance in drug development. There has also been significant interest in silencing RNA (siRNA) therapeutics, highlighting the industry's focus on novel approaches to disease treatment. With the industry focusing on cutting-edge technologies that address unmet medical needs, we feel there is little reason to fret over the size of the deals.
Impressions: 583
Artificial intelligence (AI) is changing the world and bringing
efficiencies across all industries. Pharmaceuticals is one of them. AI can
accelerate drug discovery, streamline clinical trials, and personalize
medicine. It holds the potential to revolutionize the pharma industry.It takes anywhere between 10 to 15 years and around US$ 3 billion to take a new drug from its
discovery phase to the market. AI can drastically cut both the timespan and
costs and bring life-saving yet affordable treatments to the market at a faster
pace. The global AI market in healthcare was estimated to be about US$ 21
billion in 2024 and is expected to grow to over US$ 148 billion by 2029, compounding at an annual
growth rate of 48.1 percent.Drugmakers are eager to ride the AI wave. Players like Bayer, Merck KGaA, Moderna, BMS, Roche, Astellas, Amgen, Eli Lilly, Sanofi, Novo Nordisk and Johnson & Johnson have announced
collaborations, signed deals and entered into partnerships in the AI space.
Many others, such as Exscientia, Insilico, Berg, Nimbus, Recursion, and Pharos iBio, are a step ahead and are holding clinical trials on drugs developed using AI.On its part, the US Food and Drug Administration (FDA) has
recognized the increased use of AI and machine learning (ML) and has reiterated its commitment to ensure the safety and efficacy of drugs “while facilitating innovations in their development.” To this end, the US agency
published a paper in March that lists out its
approach towards the use of AI.Novo, Lilly turn to AI for
drug discovery; AI-based firms sign multiple dealsWith increased adoption of wearable devices, e-health services and
other technology-driven offerings in medicine and healthcare, there is valuable data out there that drugmakers can analyze to get more accurate predictions of a drug’s effects on the human body. Such an analysis speeds up drug development and reduces side effects of therapies. Ergo, several large drugmakers have
signed crucial AI deals. For example, Novo Nordisk has inked a potential US$ 2.7 billion deal with Valo Health to discover and develop novel treatments for cardio-metabolic diseases. The collaboration between the two organizations will leverage the capabilities of Valo’s Opal Computational Platform, including access to real-world patient data, AI-enabled small molecule discovery and Biowire human tissue modeling platform designed to speed up the discovery and development process.Similarly, Eli Lilly has entered into a US$ 494 million deal with Fauna Bio to use the latter’s AI platform — Convergence — for preclinical drug discovery efforts in obesity. Convergence analyzes genomic data from 452 mammal species and various tissue types to
create a comprehensive dataset. By integrating human data and information from
animals with natural disease resistance, the platform can identify potential
drug targets.AI-based pharma firms like Isomorphic Labs and Biolojic are also landing multiple deals. Isomorphic is owned by Google’s parent Alphabet. Along with Google DeepMind, it has created a new AI model that can
accurately predict 76 percent of protein interactions with
small molecules. In January, Isomorphic inked deals with Novartis and Lilly for a combined
value of nearly US$ 3 billion.Biolojic Design uses computational biology and AI to transform
antibodies into programmable, intelligent medicines. Earlier this month, it
announced a multi-target drug discovery collaboration
with Merck KGaA, which includes
antibody-drug conjugates. Biolojic has also inked deals with Teva and Nektar recently.Amgen harnesses generative
biology for protein-based drugs; Sanofi ties up with OpenAIFinding a good protein drug candidate is like finding a needle in
a haystack. Drug developers typically start by looking at proteins in nature and then go through the painstaking process of shaping them into safe and effective drugs.Generative biology is a revolutionary approach to drug discovery
and development that leverages ML and AI to design novel protein therapeutics. Amgen is using generative biology to innovate new protein-based drugs that have desired structures and properties based on existing protein data inputs. In fact, the California-based multinational has said it is “integrating AI across all operational levels.” It has collaborated with PostEra to advance up to five small molecule programs.Amgen is building AI models trained to analyze one of the world’s largest human datasets on an NVIDIA full-stack data center platform, known as DGX SuperPOD, that will be installed at Amgen’s deCODE genetics’ headquarters in Reykjavik, Iceland. This system will be used to build a human diversity atlas for drug target and disease-specific biomarker discovery, providing vital diagnostics for monitoring disease progression and regression.AI can analyze individual data like genetic makeup,
lifestyle and medical history to come up with personalized therapies. Amgen is working on
AI-driven precision medicines, and potentially individualized therapies, at its
Iceland facility.Sanofi too has signed multiple deals in recent months. In May,
the French drugmaker signed a collaboration with ChatGPT maker OpenAI and Formation Bio to build AI-powered software to accelerate drug development. Sanofi is also collaborating with BioMap in a deal that could be
worth up to US$ 1 billion. The deal is expected to
enable superior prediction from limited data in a range
of therapeutic areas, including immunology, neurology, oncology, and rare
diseases.AI tools can create control
arms, digital twins and slash failure rate in clinical trialsTraditional ways of drug development are fraught with challenges,
and 90 percent of drug candidates in clinical trials tend to fail. Major reasons behind this are poor patient cohort selection and recruiting mechanisms, and the inability to monitor patients effectively during trials. AI tools like Trial Pathfinder study real data obtained from patients’ electronic health records and simulate clinical trials for the drug with different eligibility criteria. Trial Pathfinder also calculates trial hazard ratios, a scientific term that compares the survival rates of those given and not given the drug.AI and synthetic data can also be used to create control arms in
clinical trials, which can help speed up the process, reduce costs, and improve
the quality of data. For example, FDA has supported the use of a Medidata Synthetic Control
Arm in a
phase 3 trial of Medicenna’s MDNA55 in recurrent
glioblastoma.In addition to observing real-time patient data, researchers can
also create digital twins, virtual replicas of cells,
organs, or people, which they can use to simulate and predict various clinical
outcomes during a trial.Our viewThe
pharmaceutical industry is on the cusp of a revolution. In the coming years, we
hope to see some tangible results of the efforts being put in by drugmakers, AI
developers and regulatory agencies. The CPhI Annual Report 2023 has predicted
that in 2030, over half of FDA-approved drugs will involve AI in their development and/or manufacturing. The winner, in our view, will be the end-user who will have cheaper, safer and more effective treatments, delivered at a faster pace.
Impressions: 1831