FDA approvals drop 8% in 2025, with fewer blockbusters; Brinsupri, Rhapsido make it to first-in-class list
Our
update for new drug approvals by the US Food and Drug Administration (FDA) in
the first half (
Top news of 2025: Drugmakers invest in US capacities, agree to lower Medicaid prices; Pfizer buys obesity-focused biotech Metsera
The year 2025 was an eventful one, marked by increased trade
tensions, tariff threats, accelerated
Top Pharma Companies & Drugs in 2024: Merck’s Keytruda maintains top spot as Novo’s semaglutide nips at its heels
In 2024, Big Pharma players consolidated
and maintained their dominance, even as innovation continu
Molecular glue degraders: Lilly, AbbVie sign billion-dollar deals; BMS leads with three late-stage drugs
This week, we delve into molecular glue degraders (MGDs), one of the most promising frontiers in dru
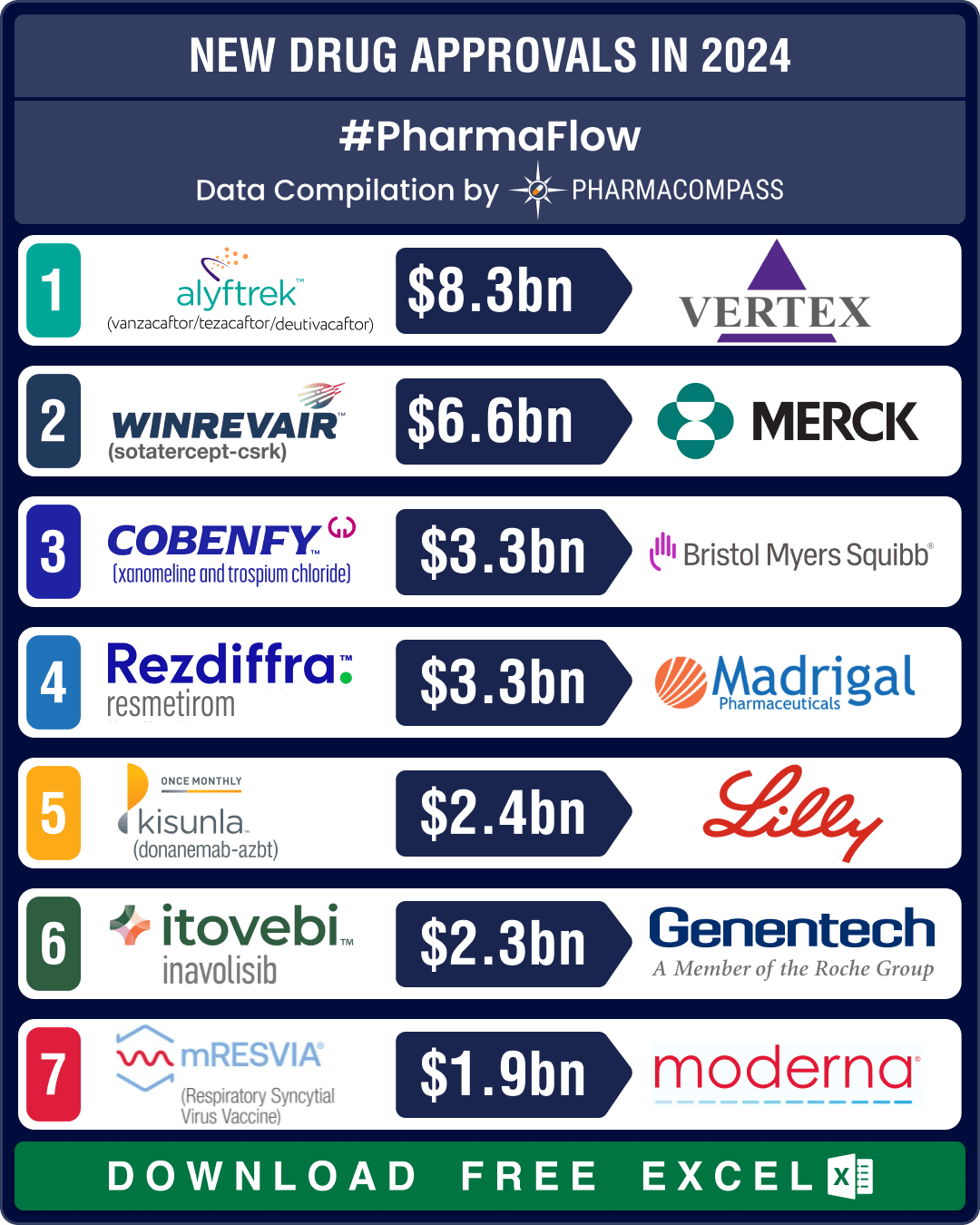
FDA okays 50 new drugs in 2024; BMS’ Cobenfy, Lilly’s Kisunla lead pack of breakthrough therapies
In 2024, the biopharma industry continued to advance on its robust trajectory of innovation. Though
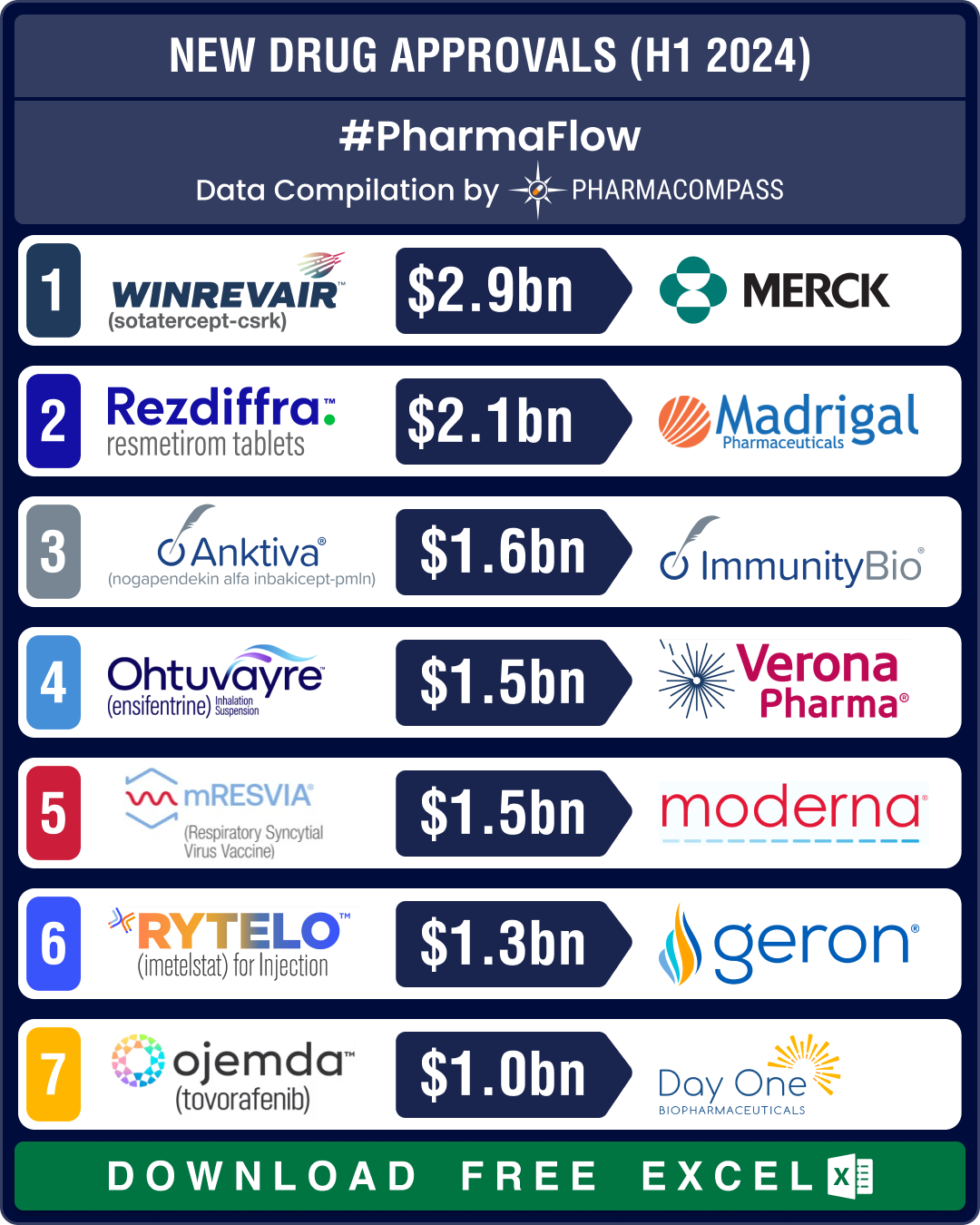
FDA approvals slump 19% in H1 2024; NASH, COPD, PAH get new treatment options
The first half of 2024 saw a significant slowdown in approvals of new drugs and biologics by the US