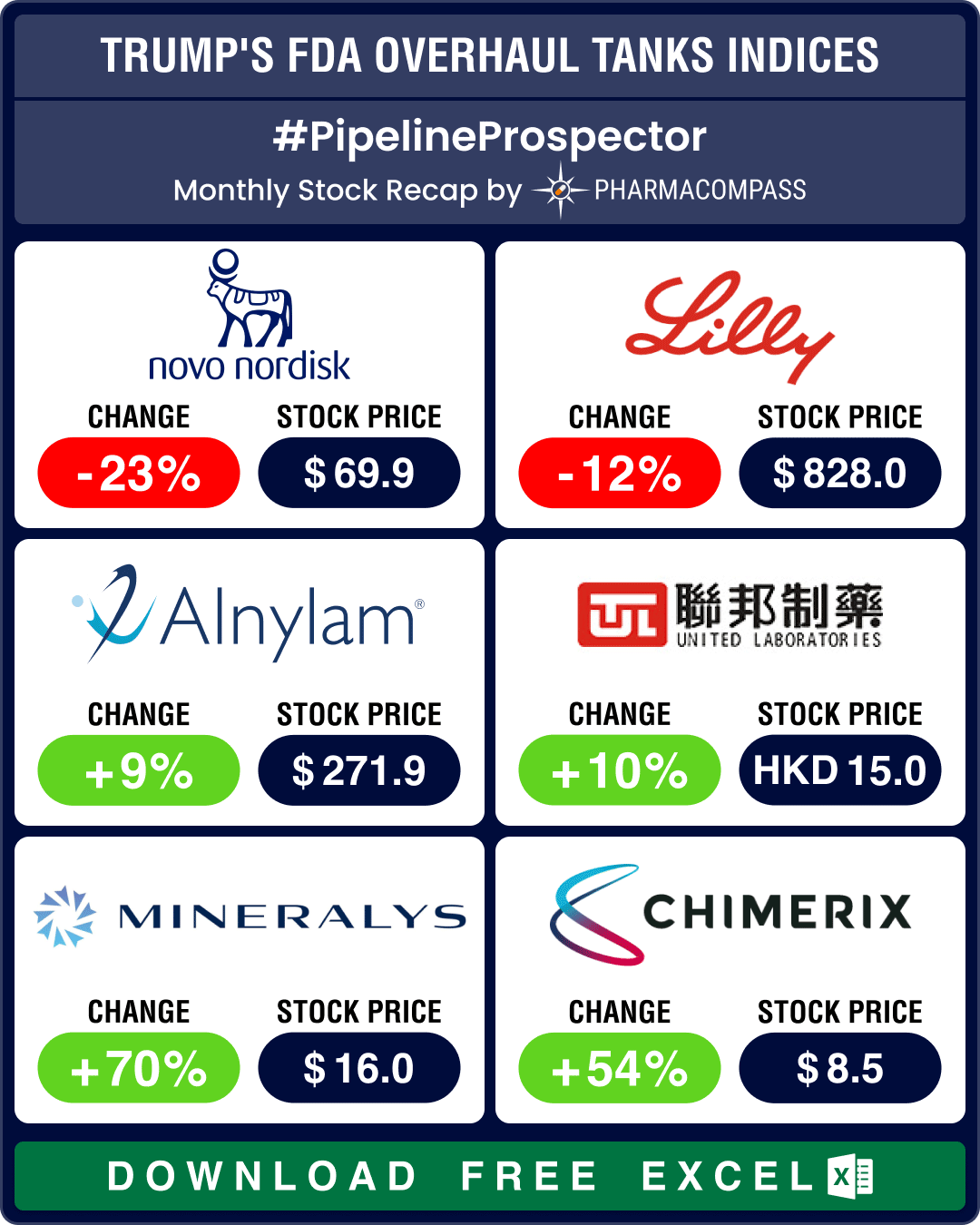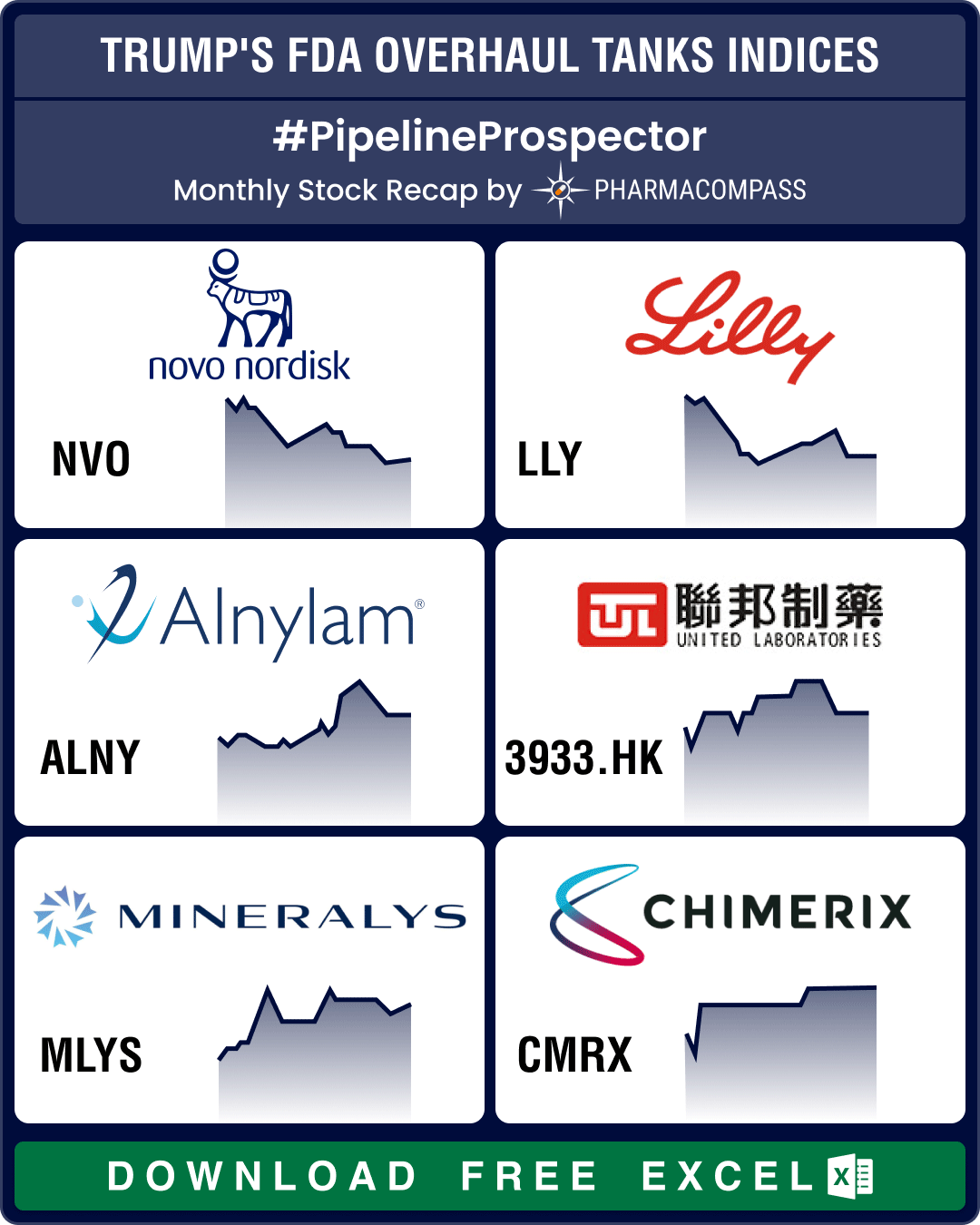
PharmaCompass has reviewed nearly 200 DMF filings available on the US Food and Drug Administration (FDA) website under the third quarter US Drug Master Files (DMFs). Our analysis shows that unlike previous quarters, where there were a number of first-to-files, this quarter the filings appear to be driven by the ‘price gouging’ debate raging on in the US for the last few months.
More players treading the Valeant path
Canada-headquartered Valeant Pharmaceuticals, which has been at the forefront of the price increase debate, has several generic challengers ready to eat into the niche markets currently monopolized by it.
Take the case of Syprine (trientine hydrochloride), a medication for Wilson’s disease acquired by Valeant in 2010. The company increased the price of Syprine from US $ 1,395 in 2013 to US $ 21,267 in 2014, generating US $ 88 million in sales. This quarter, Syprine has three DMF filings – from Watson Pharma (owned by Allergan – which merged with Pfizer earlier this week), Chemwerth Inc and Biophore India.
Nitropress, another drug acquired by Valeant and subjected to a same-day price increase of 212 percent, has also witnessed three filings. The three companies are Biophore, Emcure and Micro Labs. Interestingly, the latter two companies are currently on FDA’s import alert list. Micro Labs has also filed a DMF for the active ingredient of Isuprel – Isoproterenol Hydrochloride. Isuprel was also a victim of Valeant’s price gouging – it witnessed a same-day price increase of 525 percent in January this year.
There is more competition awaiting Valeant in other drugs, such as Elidel (pimecrolimus) – a medication for atopic dermatitis that brought in US $ 103 million in sales last year. This quarter, Concord Biotech and Biocon have filed DMFs for this drug.
Sanofi’s lawyers will be staying busy
Earlier this month, American Stock Transfer & Trust, a trustee for Genzyme Corporation rights holders, filed a lawsuit against Paris-based Sanofi. In 2011, Sanofi had acquired Genzyme Corp for more than US $ 20 billion. At the time of the acquisition, Genzyme was developing the multiple sclerosis drug – Lemtrada.
According to the lawsuit, Sanofi intentionally ignored the FDA’s concerns about Lemtrada’s clinical trial, which led to the FDA turning down Sanofi’s first drug application for Lemtrada. The suit also accused Sanofi of deliberately under-marketing the drug after its approval in November 2014. Instead, Sanofi heavily promoted a different multi-sclerosis drug, Aubagio (teriflunomide).
While it is for the court to decide whether Sanofi stalled the development of Lemtrada in favor of Aubagio to avoid paying US $ 708 million to the right-holders or not, Sanofi surely needs to get ready for considerable generic competition as Unichem Laboratories filed their DMF this quarter.
Biophore – the leading DMF filer this quarter with seven filings – continues to focus on niche molecules. Along with Dr. Reddy’s and Hetero Drugs, Biophore filed a DMF for Plerixafor. Sold as Mozobil, this Sanofi drug is used to stimulate the release of stem cells from the bone marrow into the blood stream in patients with non-Hodgkin lymphoma and multiple myeloma. This product generated Euro 69 million for Sanofi in the first half of 2015 and witnessed 21 percent growth in the United States. While Mozobil is still under patent, Sanofi’s lawyers should get ready for litigation from Indian generic companies.
More disappointments await Amgen’s anti-cancer drug
Amgen acquired Onyx Pharmaceuticals for US $ 10.4 billion in 2013 primarily to cash in on the potential of Kyprolis (carfilzomib), a cancer-treatment drug. While analysts had estimated peak sales of US $ 1.6 billion as a result of this acquisition, sales in 2014 turned out to be only US $ 331 million – a fifth of their estimates.
It seems like more disappointments await Amgen. While MSN Laboratories has filed a DMF for Carfilzomib, it seems that Amgen’s woes could be compounded by an imminent first-to-file challenge for this anti-cancer drug.
Blood thinners continue to be a favorite
In our review of the first quarter filings, Bayer’s blood thinner – Rivaroxaban – was the second-most filed product with nine DMF listings. As per our research, the focus on blood thinners, a multi-billion dollar market, continues in this quarter as well.
Bristol-Myer’s blockbuster drug Eliquis (apixaban) – an anticoagulant for the treatment of venous thromboembolic events – which generated US $ 774 million in 2014 sales (a growth of 430 percent over the previous year), had filings from leading generic players such as Dr. Reddy’s, Alembic and Zhejiang Huahai.
Our view
Given the current industry environment of ever-increasing regulations, it is critical for pharma companies to make wise product development decisions, especially when it comes to API manufacturing.
Unfortunately, the focus of the industry appears to be on price gouging, and primarily in niche segments (such as in medication for Wilson’s Disease, which is a rare genetic disorder).
In our view, this cannot be a long-term strategy since the market sizes are extremely small and with increasing compliance requirements, the returns may not be as attractive as currently envisioned.
PharmaCompass is happy to share its compilation of this quarter’s DMF filing list. Send us an email (click here) and we would be happy to help.
Also, do not forget to enquire about our Partner Company Subscription program. Under this program, PharmaCompass serves as an extension of your organization’s marketing and business development team.
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
Image Credit : The Cosmopolitan of Las Vegas by Mayer Hawthorne throws the dice for The Cosmopolitan of Las Vegas opening ceremonial 'first bet' is licensed under CC BY 2.0
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”