By PharmaCompass
2025-04-03
Impressions: 454 (Article) || 3 (Video)
The pharma industry saw its most tumultuous week in the recent past with the US Food and Drug Administration (FDA) facing an unprecedented crisis. The Trump administration’s health agency purge has left the FDA in disarray, with the Center for Biologics Evaluation and Research (CBER) Director Peter Marks forced out and mass firings causing what former commissioner Robert Califf called the end of “FDA as we've known it.”
Meanwhile, Sanofi and Alnylam celebrated a significant milestone with the FDA approving Qfitlia, the first RNA interference (RNAi) therapeutic for hemophilia A and B. While Qfitlia is the seventh hemophilia therapy to get US approval in the last three years, it is the only one indicated for both hemophilia A and B.
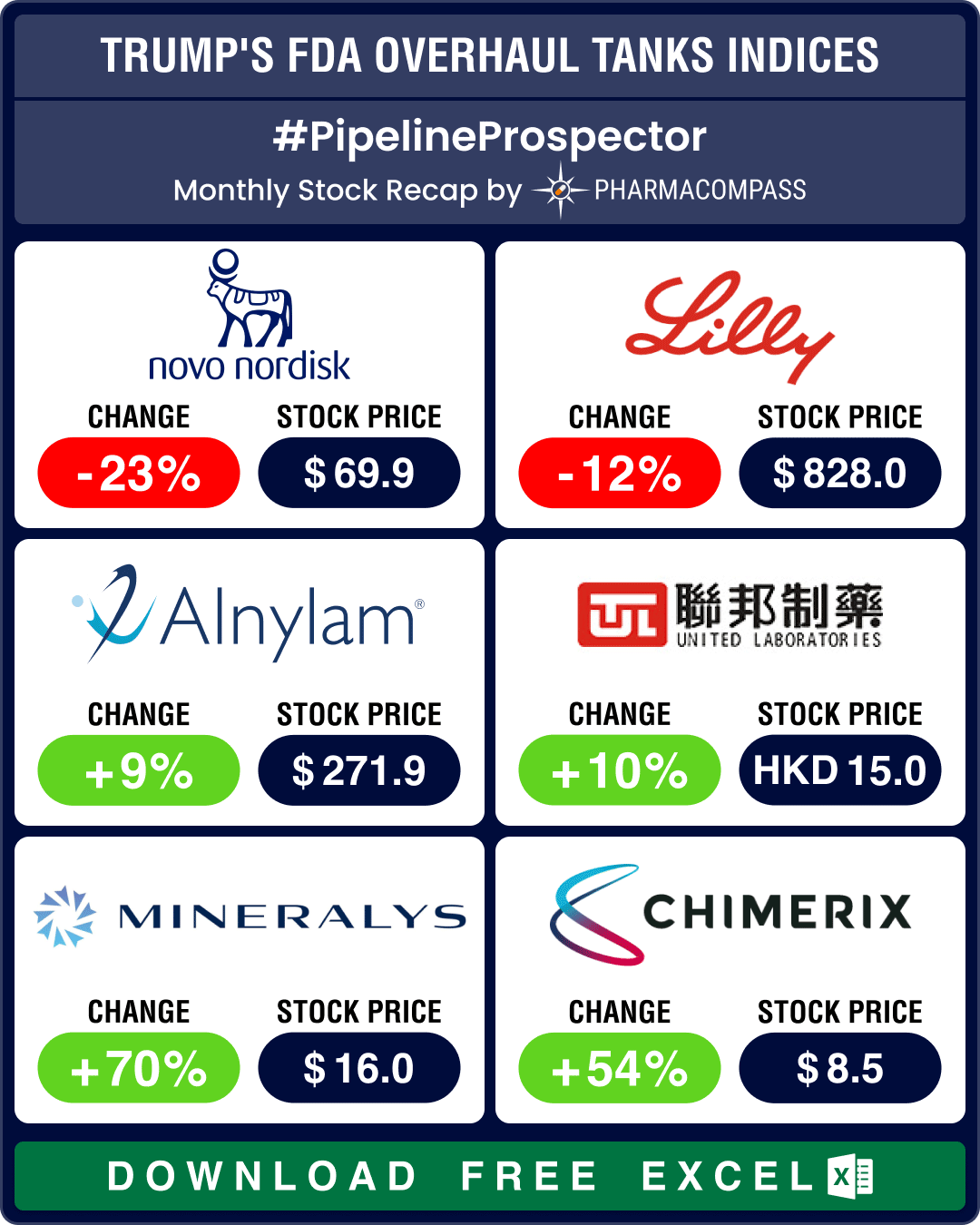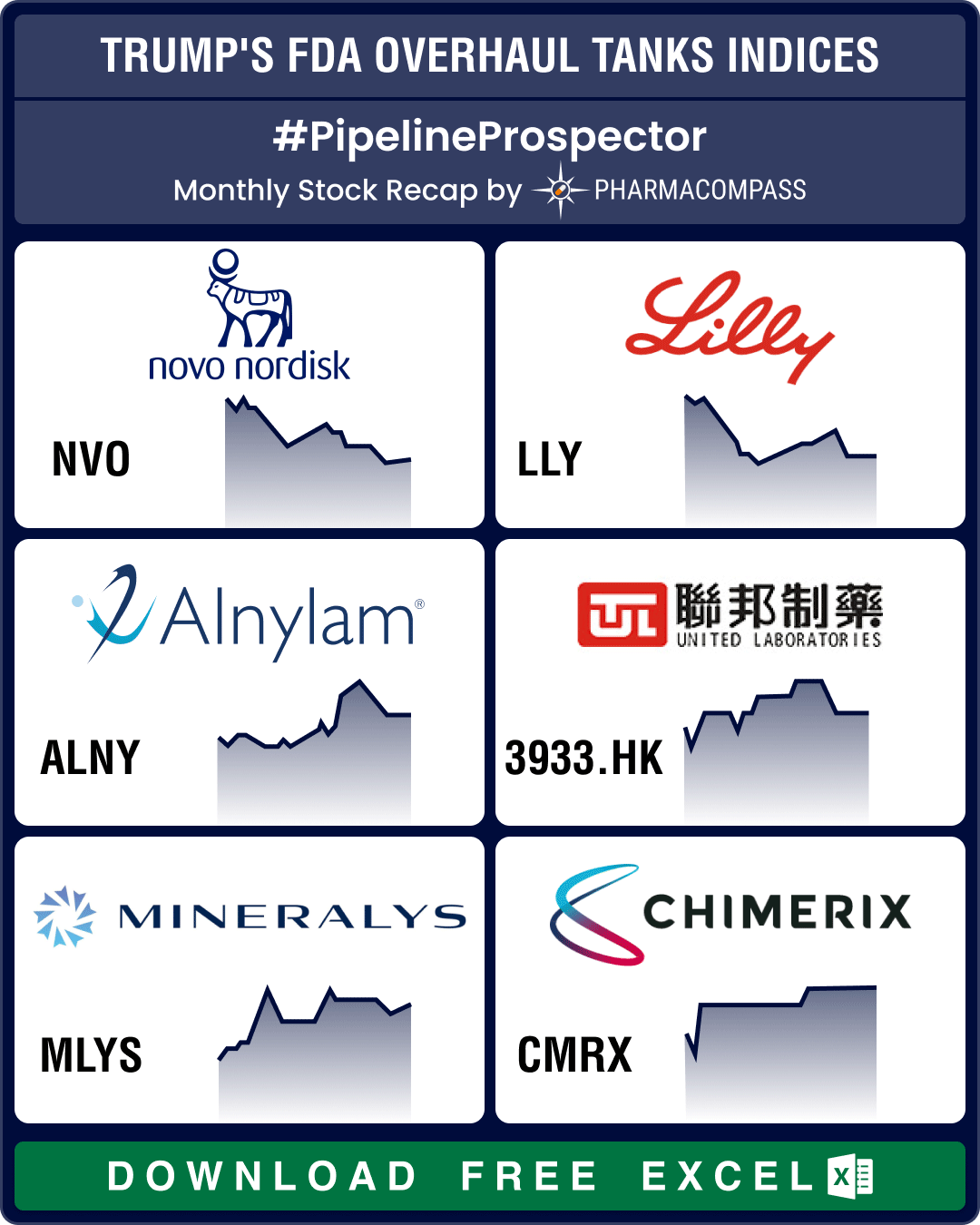
Novo Nordisk inked a deal in the obesity space for the second week in a row. Novo secured a US$ 1 billion deal with Lexicon Pharmaceuticals for its experimental oral obesity drug LX9851, which has shown promising results when combined with Novo’s glucagon-like peptide-1 (GLP-1) agonist semaglutide.
Novartis received expanded FDA approval for its prostate cancer therapy Pluvicto, now available for pre-chemotherapy use. In other regulatory news, Bavarian Nordic’s freeze-dried formulation of the Jynneos vaccine for smallpox and mpox received FDA approval, offering advantages in transportation and long-term stockpiling.
There were mixed results from clinical trials. Corcept Therapeutics reported a 30 percent reduction in disease progression risk for platinum-resistant ovarian cancer with its investigational drug relacorilant, while Merck’s Winrevair demonstrated a remarkable 76 percent reduction in mortality and morbidity risks for pulmonary arterial hypertension patients. However, Axsome Therapeutics announced plans to narrow its late-stage trial of Sunosi for major depressive disorder after the drug failed to meet its primary endpoint.
FDA leadership gutted: CBER chief forced to quit; officials find badges deactivated in mass firing
In a seismic shake-up at the FDA, Peter Marks, the director of the CBER, has been ousted from his position. This move comes amid escalating tensions between Marks and Health and Human Services (HHS) Secretary Robert F. Kennedy Jr.
RFK Jr’s controversial stance on vaccines has roiled the public health community. Marks said in his resignation letter: “It has become clear that truth and transparency are not desired by the Secretary, but rather he wishes subservient confirmation of his misinformation and lies.” Another high-profile ouster at FDA is that of Hilary Marston, the agency’s chief medical officer.
These exits are accompanied by mass layoffs across federal health agencies, with 10,000 planned job cuts underway. It turned out to be a no April Fools’ joke when FDA employees turned up to work on April 1, only to find their badges didn’t work because they no longer had a job. Former FDA Commissioner Robert Califf decried the move, saying, “FDA as we’ve known it is finished…”
The resignations and layoffs are beginning to impact the working of the agency. On Wednesday, Novavax announced that FDA had missed its deadline for making a decision on the traditional approval for its Covid-19 vaccine. Moreover, these layoffs are happening at a time when the US is dealing with a measles outbreak. The World Health Organization has issued a travel advisory and has urged people to ensure their vaccinations are up to date before traveling to the US.
Meanwhile, Tracy Beth Hoeg, a sports medicine physician who opposed key US health policies during the pandemic and questioned the use of some childhood vaccines, is now special assistant to FDA chief Martin Makary.
Sanofi-Alnylam’s Ofitlia bags FDA nod; becomes first RNAi med for hemophilia A, B
FDA has approved Qfitlia (fitusiran), marking the first RNAi therapeutic for the treatment of hemophilia A and B. Developed through a collaboration between Sanofi and Alnylam Pharmaceuticals, Qfitlia is indicated for routine prophylaxis to prevent or reduce bleeding episodes in hemophilia patients aged 12 and older.
Qfitlia is a subcutaneous therapy administered every two months, offering a less frequent dosing schedule compared to existing treatments such as Pfizer’s weekly injection Hympavzi (marstacimab-hncq) and Novo Nordisk's daily Alhemo (concizumab-mtci).
Novo inks US$ 1 bn deal with Lexicon for new type of oral obesity drug
Novo Nordisk has entered into an exclusive licensing agreement with Lexicon Pharmaceuticals to develop, manufacture, and commercialize Lexicon’s experimental obesity drug, LX9851. The deal includes upfront and near-term milestone payments of up to US$ 75 million made by Novo to Lexicon, with the total potential value reaching approximately US$ 1 billion. LX9851 is an oral medication that targets the Acyl-CoA Synthetase 5 (ACSL5) protein, which plays a significant role in fat accumulation and energy balance.
Novartis’ prostate cancer med Pluvicto wins expanded FDA nod for pre-chemo use
FDA has expanded the approved use of Novartis’ radioligand therapy, Pluvicto (lutetium Lu 177 vipivotide tetraxetan), for the treatment of prostate-specific membrane antigen (PSMA)-positive metastatic castration-resistant prostate cancer (mCRPC). This approval allows Pluvicto to be used earlier in the treatment process, prior to taxane-based chemotherapy.
Freeze-dried version of Bavarian’s Jynneos okayed: FDA has approved a freeze-dried formulation of Bavarian Nordic’s Jynneos vaccine for the prevention of smallpox and mpox (formerly known as monkeypox) in adults aged 18 and older. This new formulation offers advantages in transportation, storage conditions, and shelf life, making it more suitable for long-term stockpiling compared to the liquid-frozen version.
Corcept’s ovarian cancer drug cuts disease progression by 30% in late-stage study
Corcept Therapeutics has reported that its investigational drug, relacorilant, combined with chemotherapy, achieved the primary endpoint in a late-stage trial for platinum-resistant ovarian cancer. The combination therapy demonstrated a 30 percent reduction in disease progression risk compared to chemotherapy alone, thereby improving progression-free survival for patients.
Merck’s Winrevair slashes morbidity risks by 76%: Merck’s Winrevair (sotatercept-csrk) has demonstrated a 76 percent reduction (compared to placebo) in mortality and morbidity risks for patients with pulmonary arterial hypertension (PAH) in a phase 3 trial. The trial was halted early due to these compelling results, marking the first PAH clinical trial to be stopped for overwhelming efficacy.
Axsome narrows trial focus of MDD med: Axsome Therapeutics is narrowing the focus of a late-stage trial of Sunosi (solriamfetol) as a treatment for major depressive disorder (MDD) after an initial run of the study showed improvement only in a small subgroup of patients. The drug showed promise in alleviating excessive daytime sleepiness (EDS) among MDD patients.
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
Image Credit : Phisper Infographic by PharmaCompass license under CC BY 2.0
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”