By PharmaCompass
2022-08-11
Impressions: 1,563
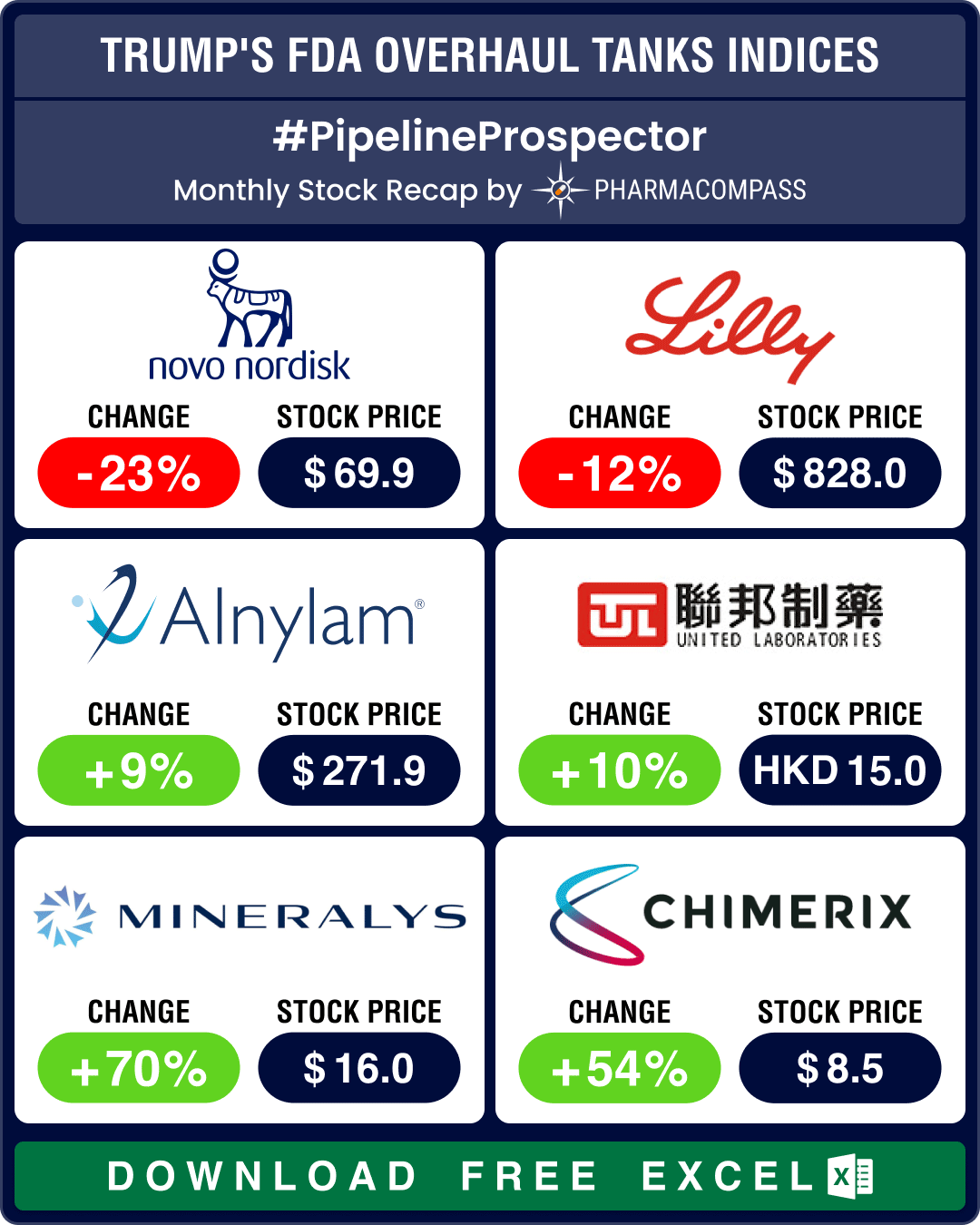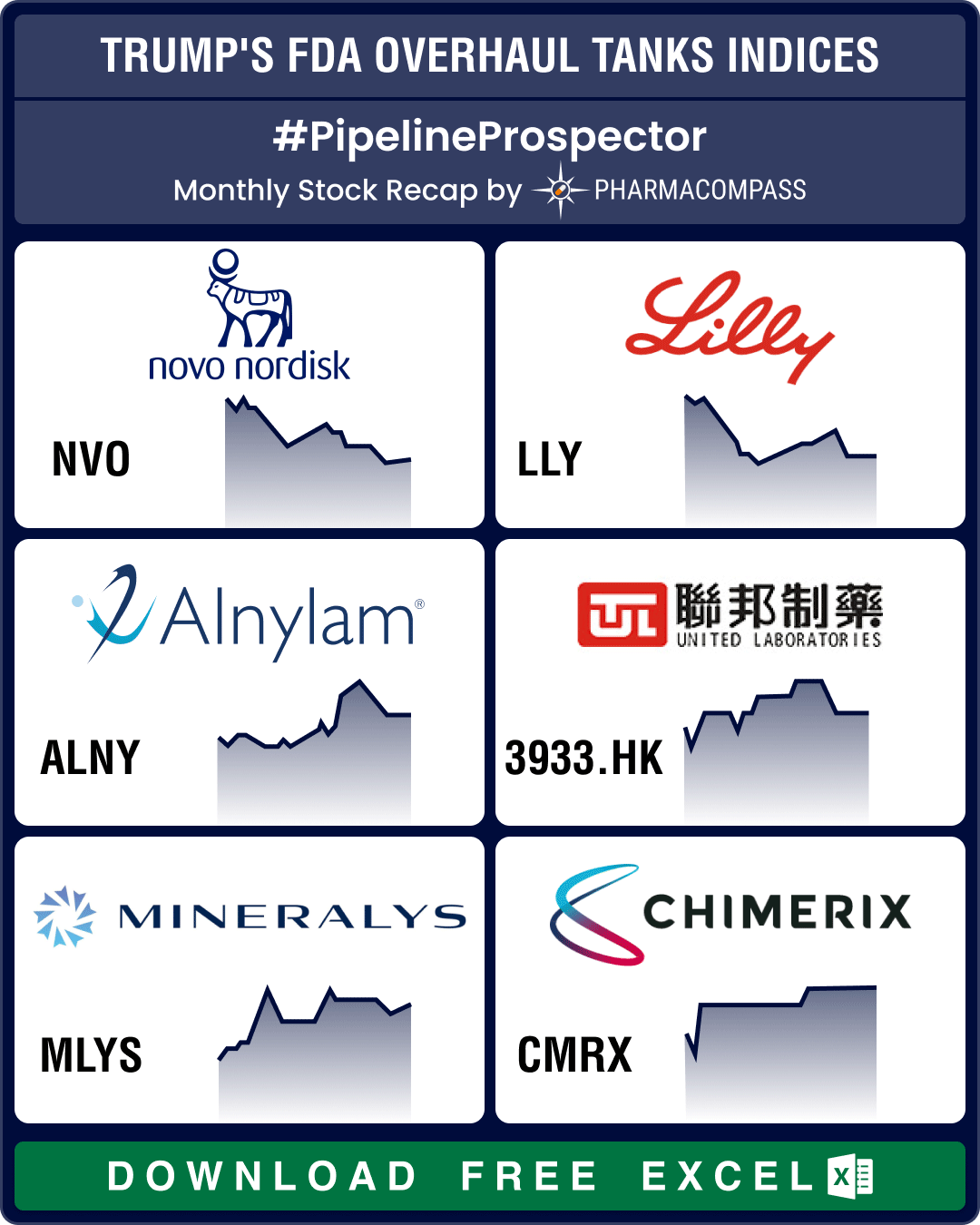
It was a big week for M&As. After a sluggish first few months, pharma companies went all out signing deals. The biggest announcement came from cash-rich Pfizer that acquired blood disorder drugmaker Global Blood Therapeutics (GBT) for US$ 5.4 billion with the intention of expanding its sickle cell disease portfolio. Amgen picked up Californian biotech ChemoCentryx for US$ 3.7 billion to help broaden its portfolio of inflammation and kidney drugs.
In other deals-related news, GSK entered into a global collaboration with Mersana Therapeutics to co-develop and commercialize the latter’s antibody drug conjugate (ADC) preclinical drug aimed at treating a range of HER-2 cancers. Gilead Sciences said it will acquire MiroBio, a privately held biotech, which is developing antibody drugs for autoimmune disorders, for US$ 405 million in an all-cash deal. And Merck has signed a potential US$ 1.1 billion research collaboration with Boston-based Cerevance to find new targets for Alzheimer's disease.
In a landmark nod, AstraZeneca and its Japanese partner Daiichi Sankyo’s Enhertu has become the first therapy to treat patients with HER2-low breast cancer in the US. The agency has also approved Pfizer and Myovant’s therapy Myfembree to treat moderate-to-severe endometriosis pain in pre-menopausal women.
Bayer’s cancer drug Nubeqa, in combination with docetaxel, has also received an FDA nod to treat patients with metastatic hormone-sensitive prostate cancer (mHSPC). Novartis and its Chinese partner BeiGene’s drug tislelizumab has met its primary goal of being as good as Bayer’s Nexavar (sorafenib) at extending the lives of patients with newly diagnosed unresectable liver cancer in a phase 3 trial.
In regulatory news, FDA has issued a Form 483 with four observations to Zydus Lifesciences’ Moraiya manufacturing plant in Ahmedabad, India. And despite finding a potential cancer-causing impurity in some samples of Merck’s blockbuster type 2 diabetes drug Januvia (sitagliptin), FDA has decided to allow Merck to distribute the drug for now, in order to avoid a shortage.
Pfizer acquires Global Blood Therapeutics for US$ 5.4 billion
Cash-rich Pfizer has acquired blood disorder drugmaker Global Blood Therapeutics (GBT) for US$ 5.4 billion. The New York-headquartered pharma said it will pay US$ 68.5 per GBT share. The buyout has added GBT’s approved drug, Oxbryta, along with two other sickle cell medicines that are in mid- to late-stage testing (including one in phase 3 clinical trials), into Pfizer’s portfolio. As of today, Pfizer has just one sickle cell disease candidate in phase 1. Pfizer expects all these blood disorder drugs to contribute US$ 3 billion in peak sales.
Oxbryta was approved in 2019 to treat sickle cell disease among patients aged 12 years or older. In December last year, the oral drug was approved for the treatment of the condition in younger patients. Oxbryta brought in US$ 195 million in 2021.
Sickle cell disease is an inherited blood disorder affecting about 100,000 people in the US. The takeover will put Pfizer shoulder-to-shoulder with Novartis, which has signaled a big push in the area. Pfizer and GBT expect the deal to close by the fourth quarter.
Terminates late-stage trial of rare heart disease drug: Pfizer has terminated a late-stage clinical trial of a rare cardiovascular disease drug candidate (PF-07265803) it had picked up as part of its US$ 11.4 billion takeover of Array BioPharma in 2019.
Drug approved to treat endometriosis pain: The FDA has approved Pfizer and Myovant’s once-a-day therapy — Myfembree — to treat moderate-to-severe endometriosis pain in pre-menopausal women for up to 24 months. A few months ago, the agency had delayed its decision and had requested additional data from the companies regarding bone mineral density. The drug was first approved in May 2021 to treat heavy menstrual bleeding associated with uterine fibroids.
Amgen picks up Californian biotech ChemoCentryx for US$ 3.7 billion
Amgen has inked a deal to acquire Californian biotech ChemoCentryx for US$ 3.7 billion to help broaden its portfolio of inflammation and kidney drugs. Amgen will pay US$ 52 in cash for each share of ChemoCentryx to seal the deal.
The deal will give the pharma access to ChemoCentryx’s potential blockbuster treatment for inflammatory disorders — Tavneos (avacopan). The drug was approved by the US Food and Drug Administration (FDA) late last year to treat a rare autoimmune disease known as anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis, in which damage to small blood vessels can lead to organ failures, especially in the kidney. In the first quarter, the drug brought in higher-than-expected sales of US$ 5.4 million in the US. Analysts have predicted over US$ 1 billion in Tavneos sales by 2027. While the drug is also approved in the EU and Japan, Amgen will only gain access to the US sales.
Along with Tavneos, ChemoCentryx has three early-stage drug candidates that target ulcerative colitis and skin conditions as well as an oral immune checkpoint inhibitor for cancer. The companies expect the deal to close in the last quarter of this year.
Lumakras+immunotherapy trial causes liver toxicity: A study of Amgen’s cancer drug Lumakras in combination with checkpoint inhibitors such as Merck’s Keytruda or Roche’s Tecentriq in KRAS G12C-mutated non-small cell lung cancer (NSCLC) helped shrink lung tumors in patients, but led to an increase in liver toxicity.
Among the 58 patients, 29 percent responded to the treatment over a median of around 18 months. While almost all trial patients treated with the highest dose of Lumakras experienced elevated liver enzyme levels, 20 percent of those given the lowest dose saw significant liver toxicity. Half the patients discontinued either Lumakras or the immuno-oncology drug due to side effects.
Amgen, Kyowa Kirin halt atopic dermatitis drug trial: Months after starting a phase 3 clinical trial of their experimental atopic dermatitis drug candidate, AMG 451, Amgen and partner Kyowa Kirin have halted patient enrollment to make changes to the study. The partners said they are changing the study to improve patient convenience and investigate a range of doses. The changes are unrelated to safety or efficacy.
AstraZeneca’s Enhertu becomes first med to treat HER2-low breast cancer
In a landmark nod, the FDA has approved AstraZeneca and its Japanese partner Daiichi Sankyo’s Enhertu as the first therapy to treat patients with HER2-low breast cancer. The approval has created a brand-new category that was once part of HER2-negative breast cancer. Around 60 percent of patients suffering from HER2-negative breast cancer fall into the HER2-low category – i.e., while they have some HER2 proteins, that’s not enough to be classified as HER2-positive.
Enhertu has been approved as a treatment for patients who have received a prior chemotherapy in the metastatic setting, or seen their cancer return within six months of completing adjuvant chemotherapy. The FDA reviewed Enhertu at an extraordinary pace by completing its assessment in just 11 days after accepting the application.
In a phase 3 study involving 550 patients, Enhertu had extended survival by more than six months and reduced the risk of disease progression or death by 50 percent and the risk of death by 36 percent in patients with previously treated HER2-low metastatic breast cancer.
GSK inks US$ 1.36 billion collaboration with Mersana for its preclinical asset
GSK has entered into a global collaboration with Mersana Therapeutics to co-develop and commercialize the US-based biotech’s antibody drug conjugate (ADC) asset, XMT-2056. The preclinical drug is aimed at treating a range of HER-2 cancers.
As part of the deal, Mersana will receive an upfront payment of US$ 100 million. It will also be eligible for up to US$ 1.36 billion in milestone payments, making the deal one of the largest deals for a preclinical ADC asset. Mersana will retain the options to co-promote and share profits in the US.
Mersana plans to launch a phase 1 trial later this year in breast, gastric and non-small cell lung cancers. In May, the drug candidate had received an orphan drug designation for gastric cancer from the FDA.
Gilead buys MiroBio for US$ 405 million: Gilead Sciences said it will acquire MiroBio, a privately held biotech which is developing antibody drugs for autoimmune disorders, for US$ 405 million in an all-cash deal. The UK-based biotech was spun out of the Oxford University three years ago. As part of the deal, Gilead will get access to MiroBio’s immune checkpoint antibody discovery platform and portfolio of immune inhibitory receptor agonists. Gilead said it will be using the platform to look for new antibodies for inflammatory diseases.
FDA issues Form 483 to Zydus Lifesciences’ Ahmedabad plant in India
The FDA has issued a Form 483 with four observations to Zydus Lifesciences’ (formerly Cadila Healthcare) Moraiya manufacturing plant in Ahmedabad, India. The agency carried out an inspection of the facility, which manufactures oral solids, injections, nasals, aerosols and transdermals, between July 26 and August 5. Zydus said none of the observations made by the FDA pertain to data integrity. The company plans to address the observations within the stipulated timeline.
CorMedix gets FDA’s CRL for antibacterial product: The FDA has issued a complete response letter (CRL) to New Jersey-based CorMedix, refusing to approve its antibacterial and antifungal product — DefenCath — until the company resolves issues surrounding its contract manufacturing organization and the supplier of the API heparin. CorMedix said the FDA recently inspected the plant of its heparin supplier and issued it a warning letter regarding its production of a different API product. CorMedix has now found an alternative contract manufacturer.
Merck inks up to US$ 1.1 billion Alzheimer’s research deal with Cerevance
Merck has signed a potential US$ 1.1 billion research collaboration with Boston-based Cerevance to find new targets for Alzheimer’s disease. Under the terms of the deal, Merck will take over a discovery-stage program and collaborate with Cerevance to develop novel targets for Alzheimer’s. The Bill Gates-backed biotech will receive an upfront payment of US$ 25 million from Merck and another potential US$ 1.1 billion in milestone payments and royalties.
FDA allows Merck to sell diabetes med Januvia despite nitrosamine impurities
A potential cancer-causing agent – Nitroso-STG-19 or NTTP – has been found in some samples of Merck’s blockbuster type 2 diabetes drug Januvia (sitagliptin). The FDA said it will temporarily allow Merck to distribute the drug containing the impurity above the acceptable intake limit to avoid a shortage. Januvia generated US$ 5.3 billion in 2021 and was ranked as Merck’s third best-selling drug behind Keytruda and Gardasil.
Bayer’s Nubeqa gets add-on nod to treat metastatic prostate cancer
The FDA has approved Bayer’s cancer drug Nubeqa, in combination with docetaxel, to treat patients with metastatic hormone-sensitive prostate cancer (mHSPC). Nubeqa (darolutamide) is already approved to treat non-metastatic castration-resistant prostate cancer in the US. Data from a phase 3 study demonstrated that adding Nubeqa to standard androgen deprivation therapy (ADT) and docetaxel chemotherapy led to a statistically significant improvement in overall survival compared to ADT plus docetaxel and placebo.
Novartis-BeiGene’s drug notches up phase 3 win in liver cancer: As Novartis and its Chinese partner BeiGene wait for a delayed FDA decision for tislelizumab in previously treated esophageal squamous cell carcinoma (ESCC), the drug has notched up another clinical victory. In a phase 3 trial, the drug met its primary goal of being as good as Bayer’s Nexavar (sorafenib) at extending the lives of patients with newly diagnosed unresectable liver cancer. BeiGene said the drug showed a consistent safety profile from previous studies involving tislelizumab and reported no new safety signals.
Karuna’s experimental schizophrenia drug: Boston-based biopharma Karuna Therapeutics said its experimental drug KarXT (xanomeline-trospium) significantly reduced symptom severity in schizophrenia patients in a phase 3 study. The experimental drug proved to be better than placebo at reducing several symptoms experienced by patients with schizophrenia in the trial.
BMS touts CAR-T therapy’s success in multiple myeloma study: Bristol Myers Squibb and partner 2Seventy Bio Inc have said their CAR-T cell therapy — Abecma — has outperformed standard drug regimens in a study on relapsed and refractory multiple myeloma. BMS said treatment with Abecma held patients’ cancer in check for a longer time as compared to standard drugs. The trial had enrolled patients who had been treated with two to four previous lines of care. The companies said they will share the full results at an upcoming medical meeting.
The PharmaCompass Newsletter – Sign Up, Stay Ahead
Feedback, help us to improve. Click here
Image Credit : Phisper Infographic by SCORR MARKETING & PharmaCompass license under CC BY 2.0
“ The article is based on the information available in public and which the author believes to be true. The author is not disseminating any information, which the author believes or knows, is confidential or in conflict with the privacy of any person. The views expressed or information supplied through this article is mere opinion and observation of the author. The author does not intend to defame, insult or, cause loss or damage to anyone, in any manner, through this article.”